-
Common NameAcibenzolar-S-methyl
-
中文通用名苯并噻二唑
-
IUPACS-methyl benzo (1,2,3) thiadiazole-7-carbothioate
-
CASS-methyl 1,2,3-benzothiadiazole-7-carbothioate
-
CAS No.135158-54-2
-
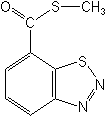
Molecular FormulaC8H6N2OS2
-
Molecular Structure
-
Category
-
ActivityPlant Activator.
This product is a selective, systemic compound, which induces host plant resistance. Acibenzolar-S-methyl has no direct effect on pathogens but acts by stimulating the plant's natural defence systems. Following application, treated plants release proteins that inhibit pathogen growth at various sites. The potential for development of resistance to this mode of action is considered low.
Actigard is recommended for the control of blue mould (Peronospora tabacina) on tobacco, downy mildew (Bremia lactucae) on lettuces, downy mildew and white rust on spinach and bacterial spot (Xanthomonas spp) and speck (Pseudomonas spp) on tomatoes. Actigard is expected to replace some of the older, higher-dose products used in these segments such as maneb and copper products.
It is recommended that preventative applications should be made early in the season to protect against bacterial and fungal pathogens. The company claims that due to the systemic activity of the product it provides more uniform protection and longer residual action than is normally achieved with fungicides that act as protectants.
The application of the water-dispersible granular formulation of this product is by ground or aerial equipment. The suspension concentrate formulation will only be used on bananas for importation to the US.
Syngenta has evaluated the potential of acibenzolar-S-methyl for the control of bacterial black spot on mangoes, bacterial black spot and angular leaf spot on cucurbits, citrus canker, black rot on Brassicae, bacterial spot on peaches and fire blight on apples.
The physiological effects of applications of acibenzolar-S-methyl to winter wheat (including foliar greening, changes in growth habit and chlorosis) have been reported in the literature (for example, see Pesticide Science, 1999).
Field trials with Actigard and A12325, a mixture of acibenzolar and trifloxystrobin were conducted on field tomatoes in Florida in 2000. Disease levels were low, but the mixture significantly reduced the incidence of early and late blight (Alternaria solani and Phytophthora infestans, respectively).
Results of trials on A12325 conducted in the USA on a range of vegetables were reported in Fungicide & Nematicide Tests (2001). These included white rust in spinach; powdery mildew in pumpkin; black rot and Alternaria leaf spot in cabbage; anthracnose, powdery and downy mildew in cucumber; powdery mildew in honeydew melon; early blight in staked tomatoes; Alternaria leaf spot in cabbage, early blight, septoria and anthracnose in tomatoes. -
CropUseCropUses:
bananas, cereals, citrus, nuts, pome fruit, rice, tobacco, tomatoes, vegetable
bananas
40 g ai/ha
cereals
30-40 g ai/ha
lettuces
52.5-70 g ai/ha
spinach
52.5 g ai/ha
tobacco/vegetables
12.5-35 g ai/ha
tomatoes
0.75-1 oz ai/100 gallons (~0.14-0.19 g ai/l)
commercial rates for other crops are still under investigation.
-
Physical PropertiesForm: Light brown solid; moderate sulfurous-like odor. Molecular weight:210.3 g/mol;Solubility at 25℃: acetone 28 g/l, dichloromethane 160 g/l, ethyl acetate 25 g/l, methanol 4.2 g/l, n-hexane 1.3 g/l, n-octanol 5.4 g/l, toluene 36 g/l.
-
Toxicology(Rat): Oral LD50 >5000 mg/kg; Inhalation LC50 (4 h) >2.79 mg/l air. (Rabbit): Dermal LD50 >2000 mg/kg.
-
Environmental ProfileHAZARDS: Fish: LC50 (96 h) 0.4 mg/l (rainbow trout), 2.8 mg/l (bluegill sunfish); (48 h) 2.4 mg/l (Daphnia). Bird: LD50 (14 d) >2000 mg/kg (mallard duck, bobwhite quail); LC50 (8 d) >5200 mg/kg (mallard duck, bobwhite quail). Bee: LD50 (48 h) (Oral) 128.3 μg/bee; (Contact) 100 μg/bee?
WATER SOLUBILITY: 7.7 mg/l (25℃)
Mallard duck
LD50 >2,000 mg/kg
Bobwhite quail
LD50 >2,000 mg/kg
Rainbow trout [96 h]
LC50 0.88 mg/L
Green algae
EC50 3.3 ppm
Daphnia magna
LC50 2.9 mg/L
Bee [48 h]
LD50 >100 μg/bee
Sheepshead minnow
LC50 1.7 ppm
Mysid shrimp
LC50 0.88 ppm
Bluegill sunfish[96 h]
LC50 1.6 mg/L
Duckweed
EC50 0.312 ppm
Earthworm
LC50 >1,000 mg/kg
Fate in :
Acibenzolar-S-methyl is practically non-toxic to terrestrial animals on an acute and subacute basis, it is practically non-toxic to insects, it is moderately to highly toxic to freshwater and estuarine aquatic animals on an acute basis and it is toxic to aquatic plants.Fate in soil:
The compound is rapidly degraded in the environment by abiotic and biotic processes. The product is rapidly degraded in aerobic soil (DT50 < 1 day), the process is slower in anaerobic systems. The major degradate is the carboxylic acid derivative, CGA-210007. This metabolite degrades rapidly in aerobic soil with a half-life of 16.5 days. Under anaerobic conditions, CGA-210007 is degraded very slowly. Light enhances the slow degradation on dry soil and the degradation of the CGA-210007 metabolite rapidly formed on wet soil.Fate in aquatic systems:
Acibenzolar-S-methyl is rapidly degraded by photolysis in aquatic systems. It has a DT50 of less than one day. The rate of degradation by hydrolysis is considerably slower; the DT50 is 3.8 years at pH 5, 23.1 weeks at pH 7 and 19.4 hours at pH 9.Fate in :
Adsorption and field studies show that movement of parent compound below the top 3 inches of soil is unlikely and the rapid degradation is the major factor in this behaviour. Acibenzolar-S-mehtyl is not expected to accumulate in aerobic sediments. The major metabolite, CGA-210007, is considerably more mobile and could accumulate in groundwater if it reaches depths at which anaerobic conditions are encountered.
Based on combined environmental exposure and ecological toxicity, it is unlikely that acibenzolar-S-methyl will pose a risk of acute or chronic toxicity to non-target animals. -
Transport InformationHazard Class:O (Obsolete as pesticide, not classified)
Porduct NewsMore
Italy approves Syngenta’s BION 50 WG
Syngenta presented Minecto Alpha for horticultural crops in Spain
Cyantraniliprole Acibenzolar-S-methyl Bronopol
EU renewed approval of acibenzolar-S-methyl

 0
0 Subscribe
Subscribe