-
Common NameTriadimefon
-
中文通用名三唑酮
-
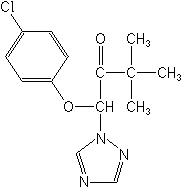
IUPAC(RS)-1-(4-chlorophenoxy)-3,3-dimethyl-1-(1H-1,2,4-triazol-1-yl)butan-2-one
-
CAS1-(4-chlorophenoxy)-3,3-dimethyl-1-(1H-1,2,4-triazol-1-yl)-2-butanone
-
CAS No.43121-43-3
-
Molecular FormulaC14H16ClN3O2
-
Molecular Structure
-
Category
-
ActivityFungicide
-
PremixTrifloxystrobin+triadimefon
Tribenuron-methyl+benzoic acid+triadimefon
Triadimefon+omethoate
Triadimefon+ethylicin
Triadimefon+carbendazim+phoxim
Imidacloprid+triadimefon
Imidacloprid+carbendazim+triadimefon
Fenvalerate+triadimefon
Chlorothalonil+prochloraz+triadimefon
Carboxin+Triadimefon
Carbendazim+triadimefon
Dry flowable, emulsifiable concentrate, granule, wettable powder. Premix Parters: chloridazon;
-
Physical PropertiesMolecular weight:293.8; Physical form:Colourless crystals, with a weak characteristic odour. Density:1.283 (21.5 °C); Composition:Racemate ( i.e. 1:1 mixture of (1R)- and (1S)- enantiomers). Melting point:Modification 1: 78 °C; modification 2: 82 °C; Vapour pressure:0.02 mPa (20 °C); 0.06 mPa (25 °C); Henry constant:9 × 10-5 Pa m3 mol-1 (20 °C); Partition coefficient(n-octanol and water):logP = 3.11; Solubility:In water 64 mg/l (20 °C). Moderately soluble in most organic solvents except aliphatics. In dichloromethane, toluene >200, isopropanol 99, hexane 6.3 (all in g/l, 20 °C).; Stability:Stable to hydrolysis; DT50 (22 °C) >1 y ( pH 3, 6, and 9).;
-
ToxicologyOral:Acute oral LD50 for rats and mice c. 1000, rabbits 250-500, dogs >500 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >5000 mg/kg. Mildly irritating to eyes and skin (rabbits). Inhalation: LC50 (4 h) for rats 3.27 mg/l air (dust), >0.5 mg/l (air). Phytotoxicity:Ornamentals may be damaged if used at excessive rates.
-
Environmental ProfileEcotoxicology:
Algae: ErC50 for Scenedesmus subspicatus 1.71 mg/l.Bees:LD50 (contact) >100 µg/bee.Birds:Acute oral LD50 for mallard ducks >4000 mg/kg. Dietary LC50 (5 d) for mallard ducks >10 000, bobwhite quail >4640 mg/kg diet.Daphnia: LC50 (48 h) 11.3 mg/l.Fish: LC50 (96 h) for bluegill sunfish 11, orfe 13.8, rainbow trout 17.4 mg/l.
Environmental fate:
Animals:In mammals, following oral administration, 83-96% is excreted unchanged in the urine and faeces within 2 to 3 d. However, metabolism occurs in the liver, mostly to triadimenol ( q.v.) and its glucuronic acid conjugates. Half-life Soil:In soil, the carbonyl group is reduced to a hydroxyl group, with the formation of triadimenol (q.v.). DT50 of triadimefon in sandy loam c. 18 d, in loam c. 6 d. Koc q.v.). -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos
ADAMA Canada moves forward with lambda-cyhalothrin sales for 2023
Mexico: Import of glyphosate drops from 16,500 tons to 8,200 tons
Glyphosate prices drop 30% in Argentina
One year extension of EU glyphosate approval failed to pass in the latest meeting
Palmer amaranth in two southern U.S. states now resistant to S-metolachlor
Related CompaniesMore
Jiangsu Sword Agrochemicals Co., Ltd.
Country: China
Triadimefon Triadimenol Tebuconazole Diniconazole Bitertanol Cyproconazole Propineb Paclobutrazol Metribuzin Bentazone
Yancheng Limin Chemical Co.,Ltd
Country: China
Triadimefon Triadimenol Hexaconazole Flutriafol Imidacloprid Paclobutrazol Tebuconazole Thifluzamide Indoxacarb Uniconazole
Shanghai Nicex International Trading Co.,Ltd.
Country: China
Oxadiazon Oxadiargyl Azoxystrobin TC Bentazone 2,4-D TC MCPA TC Nicosulfuron Tribenuron-methyl Bispyribac-sodium
Ningxia Lamtin Agricultural Development Co.Ltd.
Country: China
Oxadiargyl Bentazone Oxadiazon Metamifop Propanil
Jiangsu Kesheng Group Corporation Ltd.
Country: China
Imidacloprid Pymetrozine Pyridaben Hexythiazox Bifenthrin Chlorfluazuron Lambda-cyhalothrin Tebuconazole 2-chloro-chloromethylpyridine

 0
0 Subscribe
Subscribe