-
Common NameMethoxychlor
-
中文通用名甲氧滴滴涕
-
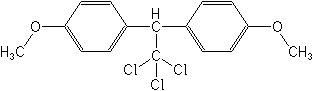
IUPAC1,1,1-trichloro-2,2-bis(4-methoxyphenyl)ethane
-
CAS1,1′-(2,2,2-trichloroethylidene)bis[4-methoxybenzene]
-
CAS No.72-43-5
-
Molecular FormulaC16H15Cl3O2
-
Molecular Structure
-
Category
-
ActivityInsecticide
-
Physical PropertiesMolecular weight:345.7; Physical form:Colourless crystals; ( tech., grey powder). Density:1.41 (25 °C); Composition:Tech. grade contains ×88% methoxychlor and ×12% related isomers. Melting point:89 °C; ( tech., 77 °C); Vapour pressure:Very low; Solubility:In water 0.1 mg/l (25 °C). Readily soluble in aromatic, chlorinated, and ketonic solvents, and vegetable oils. In chloroform and xylene 440, methanol 50 (all in g/kg, 22 °C).; Stability:Stable to oxidising agents and to u.v. irradiation. Reacts with alkalis, especially in the presence of catalytically-active metals, with loss of hydrogen chloride, but more slowly than DDT. Colour turns to pink or tan on exposure to light;
-
ToxicologyOral:Acute oral LD50 for rats 6000 mg/ kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/ kg; not a skin irritant. ADI:( JMPR) 0.1 mg/ kg b.w. [1987].
-
Environmental ProfileEcotoxicology:
Birds:Acute oral LD50 for mallard ducks >2000 mg/kg. Dietary LC50 (8 d) for bobwhite quail and ring-necked pheasants >5000 mg/ kg diet.Daphnia: LC50 (48 h) 0.00078 mg/l.Fish: LC50 (24 h) for rainbow trout 0.052, bluegill sunfish 0.067 mg/l.
Environmental fate:
Animals:Degradation in animals is principally by O-dealkylation to the corresponding phenol and diphenol, and by dehydrochlorination to 4,4'-dihydroxybenzophenone (K. S. Kapoor, J. Agric. Food Chem., 1970, 18, 1145-1152; J. Soil: DT50 in water c. 46 d. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos
ADAMA Canada moves forward with lambda-cyhalothrin sales for 2023
Mexico: Import of glyphosate drops from 16,500 tons to 8,200 tons
Glyphosate prices drop 30% in Argentina
One year extension of EU glyphosate approval failed to pass in the latest meeting
Palmer amaranth in two southern U.S. states now resistant to S-metolachlor

 0
0 Subscribe
Subscribe