-
Common NameMevinphos
-
中文通用名速灭磷
-
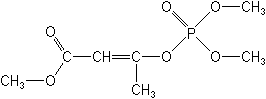
IUPAC(EZ)-2-methoxycarbonyl-1-methylvinyl dimethyl phosphate
or
methyl (EZ)-3-(dimethoxyphosphinoyloxy)but-2-enoate -
CASmethyl 3-[(dimethoxyphosphinyl)oxy]-2-butenoate
-
CAS No.7786-34-7
-
Molecular FormulaC7H13O6P
-
Molecular Structure
-
Category
-
ActivityInsecticide, acaricide
-
Physical PropertiesMolecular weight:224.1; Physical form:Colourless liquid; ( tech., pale yellow liquid). Density:1.24 (20 °C); (E)- isomer 1.235; (Z)- isomer 1.245; Composition:Tech. contains >60% m/m of the (E)- isomer and c. 20% m/m of the (Z)- isomer. Melting point:(E)- isomer 21 °C; (Z)- isomer 6.9 °C; Vapour pressure:17 mPa (20 °C); Partition coefficient(n-octanol and water):logP = 0.127; Solubility:Completely miscible with water and most organic solvents, e.g. alcohols, ketones, aromatic hydrocarbons, and chlorinated hydrocarbons. Slightly soluble in aliphatic hydrocarbons, petroleum ether, ligroin, and carbon disulfide.; Stability:Stable at ambient temperatures, but hydrolysed in aqueous alkaline solution, DT50 120 d ( pH 6), 35 d ( pH 7), 3 d ( pH 9), 1.4 h ( pH 11).;
-
Toxicology(Rat): Oral LD50 3-12 mg/kg. Inhalation LC50 9.8 mg/m3/1 hr. Dermal LD50 51-60 mg/kg. For eyes and skin, not an irritant at very low, nontoxic doses
-
Environmental ProfileEcotoxicology:
Bees:Toxic to bees; LD50 0.027 mg/bee.Birds:Acute oral LD50 for mallard ducks 4.63, chickens 7.52, pheasants 1.37 mg/ kg.Fish: LC50 (48 h) for rainbow trout 0.017, bluegill sunfish 0.037 mg/l.
Environmental fate:
Animals:In mammals, following oral administration, elimination occurs in 3-4 days in the form of metabolites in the urine and faeces.Plant:In plants, hydrolysed rapidly to less toxic products, including phosphoric acid dimethyl ester and phosphoric acid. The (E)- isomer is more rapidly degraded than the (Z)- isomer. Metabolism and degradation have been reviewed (K. I. Beyno -
Transport InformationSignal Word:DANGER; Hazard Class:Ia(Extremely hazardous)
Porduct NewsMore
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos
ADAMA Canada moves forward with lambda-cyhalothrin sales for 2023
Mexico: Import of glyphosate drops from 16,500 tons to 8,200 tons
Glyphosate prices drop 30% in Argentina
One year extension of EU glyphosate approval failed to pass in the latest meeting
Palmer amaranth in two southern U.S. states now resistant to S-metolachlor

 0
0 Subscribe
Subscribe