-
Common NameDisulfoton
-
中文通用名乙拌磷
-
IUPACO,O-diethyl S-2-ethylthioethyl phosphorodithioate
-
CASO,O-diethyl S-[2-(ethylthio)ethyl] phosphorodithioate
-
CAS No.298-04-4
-
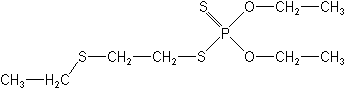
Molecular FormulaC8H19O2PS3
-
Molecular Structure
-
Category
-
ActivityAcaricide/Miticide, insecticide
-
PremixDry seed dressing, emulsifiable concentrate, granule. Premix Parters: ametryn; amitrole; amitrole ammonium thiocyanate; amitrole ammonium thiocyanate ethidimuron; amitrole bromacil; amitrole dichlorprop; amitrole ethidimuron; amitrole simazine; bromacil; bromacil sodium chlorate sodium metaborate; bromacil terbutryn; 2,4-D MSMA; fluometuron; glyphosate; glyphosate simazine; hexazinone; imazapyr; MCPA; MSMA; oryzalin; paraquat; sodium chlorate sodium metaborate; thidiazuron.
-
Physical PropertiesMolecular weight:274.4; Physical form:Colourless oil, with a characteristic odour; ( tech., pale yellow oil). Density:1.144 (20 °C); Melting point:<-25 °C; Boiling point 128°C at 1.3 hPa; Flash point:133 °C ( tech.); Vapour pressure:7.2 mPa (20 °C); 13 mPa (25 °C); 22 mPa (30 °C); Henry constant:0.24 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 3.95; Solubility:In water 25 mg/l (20 °C). Readily miscible with n-hexane, dichloromethane, isopropanol, toluene. Stability:Stable under normal storage conditions. Relatively stable in acidic and neutral media. Hydrolysed in alkaline media: DT50 (22 °C) 133 d ( pH 4), 169 d ( pH 7), 131 d ( pH 9). Miscible in n-hexane, dichloromethane, 2-propanol, toluene. Stable in normal storage.
-
ToxicologyOral:Acute oral
LD50 for male and female rats 2-12, male and female mice 7.5, female dogs c. 5 mg/kg. Percutaneous:Acute percutaneous LD50 for male rats 15.9, female rats 3.6 mg/kg. Not irritating to skin and eyes (rabbits). Inhalation: LC50 (4 h) for male rats c. 0.06, female rats c. 0.015 mg/l (for aerosol). Phytotoxicity:Non-phytotoxic when used as di -
Environmental ProfileEcotoxicology:
Bees:Toxic to bees, depending on the mode of application.Birds:Acute oralLD50 for bobwhite quail 39 mg/kg. Dietary LC50 (5 d) for mallard ducks 692, bobwhite quail 544 mg/kg diet.Daphnia: LC50 (48 h) 0.013-0.064 mg/l.Fish: LC50 (96 h) for bluegill sunfish 0.039, rainbow trout 3 mg/l.
Environmental fate:
Animals:In rats, following oral administration, 14C-disulfoton is rapidly absorbed, metabolised, and the radioactivity excreted in the urine. The main metabolites are disulfoton sulfoxide and sulfone, their corresponding oxygen analogues and diethylthiSoil:In soil, disulfoton is very rapidly degraded. The metabolism is similar to that in animals and plants. It exhibits medium to low mobility in soil.Plant:In plants, disulfoton is very rapidly metabolised. The metabolism is the same as in animals.
WATER SOLUBILITY: 25 mg/l at 20° C. -
Transport InformationSignal Word:DANGER; Hazard Class:Ia (Extremely hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe