-
Common NameFlorasulam
-
中文通用名双氟磺草胺
-
IUPAC2′,6′,8-trifluoro-5-methoxy[1,2,4]triazolo[1,5-c]pyrimidine-2-sulfonanilide
-
CASN-(2,6-difluorophenyl)-8-fluoro-5-methoxy[1,2,4]triazolo[1,5-c]pyrimidine-2-sulfonamide
-
CAS No.145701-23-1
-
Molecular FormulaC12H8F3N5O3S
-
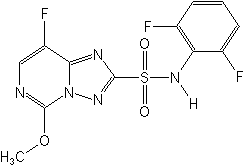
Molecular Structure
-
Category
-
ActivityFlorasulam is a post-emergence herbicide for the control of broadleaf weeds in cereals. It can be applied from the 4th leaf stage of wheat until the flag leaf stage but Dow recommends that it is applied from the end of tillering until the ear measures 1 cm (crop 21-30 cm tall). The company notes that the control of Galium aparine is not diminished by late application. Dow reports the product is active over a wider temperature range than competitors and is ideally positioned for late winter / early spring treatments when temperatures begin to exceed 5°C. Florasulam can be tank-mixed with other herbicides, with fungicides and with liquid fertilisers. In field trials, Dow has demonstrated that application rates can be reduced when the herbicide is tank-mixed with liquid fertilisers.
Root uptake studies concluded that seven times more florasulam was taken into Galium aparine roots than wheat. After 48 hours, there was ten times more material in the aerial parts in the weed than the crop. The translocated material was located in the stems and axial meristems. Foliar uptake studies indicated a similar pattern with greater translocation in the weed. Movement appears to occur in both xylem and phloem. The principal pathway for metabolism in wheat is hydroxylation at the 4-position of the anilide ring followed by glucose conjugation. The half-life of florasulam in wheat is 3.5 hours compared to 33 hours in susceptible species (Galeopsis tetrahit, Polygonum lapathifolium).
Because of its short half-life, florasulam is safe to rotational crops. In 2003, UK approval was obtained for the plantback of vegetables including brassicas in the same year that Boxer-treated crops are harvested. Carrots may be planted the following calendar year.
Field trials were conducted in Europe in spring 1999 and spring 2000 with two formulations of florasulam + fluroxypyr to investigate the control of key cereal weeds under different conditions. GF-184 (2.5 g ai/L florasulam + 100 g ai/L fluroxypyr) and GF-185 (1.0 g ai/L florasulam + 100 g ai/L fluroxypyr) were applied at early- and late-spring timings. GF-184 provided good control of Galium aparine and Matricariachamomilla at 1.5 L/ha whereas GF-185 required 1.8 L/ha to achieve a satisfactory level of control of Galium in early applications. Both formulations also provided good control of Polygonum spp, Sinapis arvensis, Stellaria media, Epilobium spp and Brassica napus. GF-184, with the higher dose of florasulam, provides stable control of the key weeds at the early timing when soil temperatures are fluctuating and it can be used up to the flag stage of the crop. This formulation should allow earlier applications than with fluroxypyr alone which requires stable soil temperatures of 4 °C or greater and will provide a broader spectrum of weed control. GF-185 is best suited to a later timing.
Trials conducted in 2000/2001 indicated a potential for the use of florasulam in newly-established hedgerows and forest nursery beds (see BCPC 2001, p733). Applications of florasulam at 15 g ai/ha, made 2 months after sapling transplantation proved selective in hawthorn, blackthorn, dog rose, grey willow, white poplar, silver birch, Norway spruce and hazel.
Dow AgroSciences is developing the product for use on new-laid lawns and pasture and investigating the possibility of developing products for nursery crops. For use on onions, the product should be applied pre-emergence or incorporated prior to planting. Field trials conducted in Lolium perenne, L multiflorum, Agrostis stolonifera, Festuca rubra rubra and Koeleria macrantha showed that florasulam and its mixture with fluroxypyr were highly selective in these seed grass species; the treatments did not adversely affect seed production.
Dow reports that the florasulam + fluroxypyr formulations, containing two actives with different modes of action, will help to minimise the emergence of herbicide resistance. -
CropUseCropUses:
Barley, oats, rye, spelt, triticale, wheat
Small-grain cereals
5 g ai/ha
-
PremixMCPA-sodium+Florasulam
MCPA-sodium+Carfentrazone-ethyl+Florasulam
MCPA-isooctyl+Mesosulfuron-methyl+Florasulam
MCPA-isooctyl+Florasulam+Carfentrazone-ethyl
ISOXABEN+FLORASULAM
Fluroxypyr-meptyl+Carfentrazone-ethyl+Florasulam
Fluroxypyr+florasulam
Florasulam+Tribenuron-methyl
Florasulam+Mesosulfuron-methyl
Florasulam+MCPA-isooctyl+fluroxypyr-meptyl
Florasulam+MCPA-isooctyl
Florasulam+MCPA ester
Florasulam+Halosulfuron-methyl
Florasulam+Halauxifen-methyl
Florasulam+Fluroxypyr-meptyl
florasulam+fluroxypyr+MCPA
Florasulam+flumetsulam
Florasulam+Flucarbazone-Na
Florasulam+Clodinafop-propargyl
Florasulam+Amidosulfuron
Florasulam+2,4-D-ethylhexyl
florasulam+2,4-d 2-ethylhexyl ester
Carfentrazone-ethyl+Florasulam
2,4-D-ethylhexyl+Clodinafop-propargyl+Florasulam
2,4-D-2-ethylhexyl+florasulam
-
Physical PropertiesMolecular weight:359.3; Melting point:193.5-230.5 °C; Vapour pressure:1 × 10-2 mPa (25 °C); Partition coefficient(n-octanol and water):logP = -1.22 ( pH 7.0); pKa:4.54; Solubility:In water 6.36 g/l ( pH 7.0, 20 °C).;
-
Environmental Profile
Ecotoxicology:
Algae: EC50 (72 h) for algae 8.94 mg/l.Bees: LD50 (48 h, oral and contact) >100 mg/bee.Birds:Acute oral LD50 for quail 1046 mg/kg. Dietary LC50 for quail and mallard ducks >5000 mg/ kg diet.Daphnia: LC50 (48 h) >292 mg/l.Fish: LC50 (96 h) for bluegill sunfish >98, rainbow trout >96 mg/l.Worms: LC50 (14 d) for earthworms >1320 mg/ kg.
Environmental fate:
Fate in soil:
In the laboratory, florasulam had a soil half-life of <1 - 4.5 days; in the field it had a soil half-life of 2-18 days. Results published by Dow in the Journal of Agrichemical Food demonstrated that this half life is dependent on temperature and independent of moisture levels. Dow reported the half-life degradation of florasulam in soil to range from 1.0 to 8.5 days at 20 to 25 °C and from 6.4 to 85 days at 5 °C.Fate in aquatic systems:
In natural systems, florasulam has a half-life of 9-29 days in the dark. The anaerobic aquatic degradation DT50 is 13 days; the aerobic DT50 is 3 days. Photolysis is also an important method of degradation. Florasulam's photolytic half-life is approximately five days.
The EU SCP concluded that two metabolites of florasulam posed no risk to non-target aquatic organisms and would not exceed the European threshold for groundwater contamination. -
Transport InformationHazard Class:O (Obsolete as pesticide, not classified)
Porduct NewsMore
ADAMA launches Timeline® FX, to deliver superior control and flexibility over broadleaf weeds
Pinoxaden Florasulam Fluroxypyr
ICL launches new selective herbicide Esteron T for turf areas
Dow AgroSciences received approval for herbicide Zypar™ in Denmark
Adama Canada: new Outshine broadleaf herbicide for cereals
China approves Dow's herbicide Florasulam•MCPA
Related CompaniesMore
Shandong Sino-Agri United Biotechnology Co., Ltd.
Country: China
Nitenpyram Pyridaben Imidacloprid Acetamiprid Dicamba Thiacloprid
Zhangye Dagong Pesticide Chemistry Co., Ltd.
Country: China
Mesotrione Flumetralin Brassinolide Butralin Trifluralin Glyphosate
Country: China
MCPA Cyproconazole Imazamox Flumetsulam Haloxyfop-P-methyl Penoxsulam Florasulam Prothioconazole Clothianidin
Jiangsu Good Harvest-Weien Agrochemical Co., Ltd.
Country: China
Glyphosate S-metolachlor Metaldehyde Metamitron Penoxsulam Florasulam Mesotrione Amicarbazone Pyraclostrobin Mesosulfuron-methyl
Shenzhen Baocheng Chemical Industry CO.,LTD.
Country: China
Glyphosate Emamectin benzoate Lambda-cyhalothrin Pyraclostrobin Bromacil Glufosinate-ammonium Tembotrione Prothioconazole Topramezone Chlorantraniliprole

 0
0 Subscribe
Subscribe