-
Common NameFuralaxyl
-
中文通用名呋霜灵
-
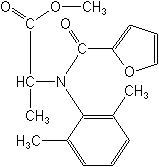
IUPACmethyl N-(2-furoyl)-N-(2,6-xylyl)-DL-alaninate
-
CASmethyl N-(2,6-dimethylphenyl)-N-(2-furanylcarbonyl)-DL-alaninate
-
CAS No.57646-30-7
-
Molecular FormulaC17H19NO4
-
Molecular Structure
-
Category
-
ActivityFungicide
-
Physical PropertiesMolecular weight:301.3; Physical form:White, odourless crystals. Density:1.22 (20 °C); Melting point:70 °C and 84 °C (dimorphic); Vapour pressure:0.07 mPa (20 °C); Henry constant:9.3 × 10-5 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 2.7; Solubility:In water 230 mg/l (20 °C). In dichloromethane 600, acetone 520, methanol 500, benzene 480, hexane 4 (all in g/ kg, 20 °C).; Stability:Relatively stable in neutral or weakly acidic media. Less stable in alkaline media; on hydrolysis, DT50 (20 °C) ( calc.) >200 d at pH 1 and pH 9, 22 d at pH 10. S;
-
ToxicologyOral:Acute oral LD50 for rats 940, mice 603 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats >3100, rabbits 5508 mg/kg. Weak skin and eye irritant (rabbits). Non-sensitising to skin (guinea pigs). ADI:0.006 mg/ kg b.w. (Bayer, 1996).
-
Environmental ProfileEcotoxicology:
Algae: EC50 for Scenedesmus subspicatus 27 mg/l.Bees:Not toxic to bees when used as directed; LD50 (24 h, oral) 5 - >20 mg/bee.Birds:Acute oral LD50 (8 d) for penguin ducks and Japanese quail >6000 mg/kg. LC50 (8 d) for Japanese quail >6000 ppm.Daphnia: LC50 (48 h) 39.0 mg/l.Fish: LC50 (96 h) for rainbow trout 32.5, crucian carp 38.4, guppy 8.7, catfish 60.0 mg/l.Worms: LC50 (14 d) for earthworms 510 mg/ kg.
Environmental fate:
Animals:After oral administration, furalaxyl is rapidly metabolised and eliminated via urine and faeces. The metabolic pathways are independent of sex or dose level administered. Residues in tissues were generally low, and there was no evidence for accumulation oSoil:In soil, the compound dissipated with DT50 31-65 d (20-25 °C). Degradation is by cleavage of the ester bond and by N-dealkylation.Plant:Furalaxyl is metabolised to polar, water-soluble, partially acidic, probably conjugated, degradation products. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe