-
Common NameS-metolachlor
-
中文通用名精异丙甲草胺
-
IUPACmixture of 80–100% 2-chloro-N-(6-ethyl-o-tolyl)-N-[(1S)-2-methoxy-1-methylethyl]acetamide and 20–0% 2-chloro-N-(6-ethyl-o-tolyl)-N-[(1R)-2-methoxy-1-methylethyl]acetamide
or
mixture of 80–100% (aRS,1S)-2-chloro-6′-ethyl-N-(2-methoxy-1-methylethyl)acet-o-toluidide and 20–0% (aRS,1R)-2-chloro-6′-ethyl-N-(2-methoxy-1-methylethyl)acet-o-toluidide -
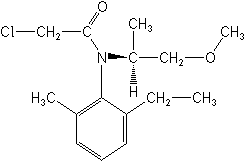
CAS2-chloro-N-(2-ethyl-6-methylphenyl)-N-[(1S)-2-methoxy-1-methylethyl]acetamide
-
CAS No.87392-12-9
-
Molecular FormulaC15H22ClNO2
-
Molecular Structure
-
Category
-
ActivityThe major contributor to herbicide activity in S-metolachlor is the pair of 1S-isomers.
Absorption of S-metolachlor is principally via the emerging shoot (coleoptile of grasses) though there is also some uptake by the roots. Herbicidal activity is expressed as failure of sensitive species to emerge and tightly-furled leaves in grass weeds which do emerge following treatment. Established weeds are much less susceptible to metolachlor. Although grass weeds are more susceptible, S-metolachlor can provide control of certain small-seeded broadleaved weeds. Herbicidal activity is attributed to inhibition of cell division and elongation, due to blockage of protein synthesis associated with root growth and inhibitory effects on the synthesis of lipids and giberellins.
Trials in Australia showed that S-metolachlor applied at high rates provides useful suppression of ryegrass (Lolium spp) in barley and oats and this has now been added to the label in Australia. Further trials are in progress to study the safety in wheat. Dual Gold has been shown to provide good control of toad rush (Juncus bufonius) in a range of Australian crops.
S-Metolachlor has a wide application window and can be used pre-plant incorporated, early pre-plant, pre-plant, pre-emergence and early post-emergence. Post-emergence applications should be made before grasses exceed the 2 leaf stage. The product provides 4-6 weeks residual activity against newly emerging weeds.
Safener technology is available for use with some products containing S-metolachlor; benoxacor is used to protect maize from damage under normal use and in adverse conditions. Seed treatment with safeners such as fluxofenim (Concep III) allows S-metolachlor-based products to be used in Sorghum bicolor to control Sorghum halapense (johnsongrass).
Since the launch of metolachlor in the mid-70s, there has been only one report of (weak) weed cross-resistance in Lolium rigidum in Australia.
Herbicide -
CropUseCropUses:
Asparagus, barley, beans (dry, succulent), brassicas, carrots, celery, corn (field, sweet), cotton, cucurbits, grass seed, horseradish, maize, oats, oilseed rape, onion (dry, green), pasture, peach, peanuts, pod crops, potatoes, pulses, sorghum, soybeans, sugar beet, sugar cane, sunflowers, tobacco, triticale, vegetables, wheat
Maize
1.0-2.0 kg ai/ha (depending on the organic material content of the soil)
-
PremixSulfentrazone+S-metolachlor
S-metolachlor+Terbuthylazine
Oxyfluorfen+S-metolachlor
Nicosulfuron+S-metolachlor+Atrazine
Atrazine+S-metolachlor
Atrazine+mesotrione+S-metolachlor
Atrazine Bicyclopyrone Mesotrione S-metolachlor
Emulsifiable concentrate, granules, smoke pellet, suspension concentrate. Premix Parters: acetochlor; chlorimuron-ethyl; flufenacet; isoproturon; metolachlor; S-metolachlor; sulfentrazone;
Type
AI concn
Emulsifiable concentrate (EC)
960 g ai/L
Granule (G)
25% ai
Suspo-emulsion (FW, SC)
290-400 g/L (in various combinations with triazines)
-
Physical PropertiesMolecular weight:283.8; Physical form:Clear yellow to brownish liquid, with an unspecific odour. Density:1.117 (20 °C); Composition:A mixture of isomers in the proportion 80-100% (S)- to 20-0% (R)- Melting point:-61.1 °C; Flash point:190 °C (EEC A9); Vapour pressure:3.7 mPa (25 °C); Partition coefficient(n-octanol and water):logP = 3.05 (20 °C); Solubility:In water 480 mg/l (25 °C); Stability:Hydrolysis DT50 >200 d ( pH 7-9, 20 °C).;
-
ToxicologyOral:Acute oral LD50 for rats 2672 mg/kg. Percutaneous:Acute dermal LD50 for rabbits >2000 mg/kg; non-irritant to skin and eyes (rabbits); may cause sensitisation by skin contact (guinea pigs). Inhalation: LC50 (4 h) for rats >2910 mg/m3. Phytotoxicity:Tolerated by most broad-leaved crops, maize, sorghum (when safened wth fluxofenim or oxabetrinil). ADI:0.1 mg/kg b.w.
-
Environmental ProfileEcotoxicology:
?Algae: EC50 (72 h) 0.006-0.14 mg/l.Bees: LD50 (oral) >0.085 mg/bee; (contact) >0.2 mg/bee.Birds:Acute oral LD50 for bobwhite quail and mallard ducks >2510 mg/kg. Dietary LC50 (8 d) for bobwhite quail and mallard ducks >5620 ppm.Daphnia: LC50 (48 h) 11.2 mg/l.Fish: LC50 (96 h) for rainbow trout 1.2, bluegill sunfish 3.2 mg/l.Worms: LC50 (14 d) for earthworms 570 mg/kg soil.?
Environmental fate:?
Animals:Rapidly oxidised by rat liver oxygenases via dechlorination, O-demethylation and side-chain oxidation; conjugation by glutathione S-transferases.Soil:Major aerobic metabolites are derivatives of oxanilic and sulfonic acids; DT50 (field) 11-30 d; DT90 (field) 36-90 d.Plant:Metabolism involves dechlorination and conjugation to glutathione S-transferases, followed by further degradation to polar, water-soluble, non-volatile metabolites.
Mallard duck
LD50 >2500 mg/kg
Bobwhite quail
LD50 >2500 mg/kg
Rainbow trout [96 h]
LC50 (96h) 1.2 mg/L
Daphnia [48 h]
LC50 26 mg/L
Earthworm
LC50 570 ppm in soil
Bees
non-toxic
Fate in soil:
Breakdown is largely microbial and hence faster in warm, moist soils. Microbial degradation occurs in aerobic and, to a lesser degree, in anaerobic soils.
Volatilisation from soil is negligible.
Leaching potential is slight to moderate depending on the soil type with clay and organic matter being key factors in determining the mobility. Koc values vary from 110-369. The rapid field dissipation and lower dose rate of S-metolachlor mean that the risk of groundwater contamination is significantly reduced compared to the racemic active ingredient, metolachlor.
A modelling study using the PELMO program, using the Focus criteria for maize, indicated that the risk of groundwater contamination following annual applications of S-metolachlor in Europe are very low and unlikely to exceed the EU trigger value of 0.1 μg/L. This finding was corroborated by the results of a 3-year field lysimeter study.
DT50 (20°C) = 7-53 days (median 14 days)
DT50 = 11 days (sandy loam, Switzerland)
DT50 = 25 days (sandy loam, Switzerland)
DT50 = 18 days (silty loam, Switzerland)
DT50 = 30 days (silty clay loam, France)
DT50 = 30 days (loam, France)Fate in Water:
Hydrolysis half-life > 200 days at pH 1-9.
Slow photolysis rate; <10% degradation after 30 days exposure.
Aquatic degradation under outdoor conditions; DT50 = 15-50 days. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Palmer amaranth in two southern U.S. states now resistant to S-metolachlor
Syngenta Portugal presents Camix herbicide
UPL launches first mixture of Glufosinate and S-metolachlor herbicide in Brazil
Glufosinate-ammonium S-metolachlor
Canada approved UPL’s KOMODO Herbicide
Syngenta launches Acuron Flexi herbicide in Canada
Bicyclopyrone Mesotrione S-metolachlor
DuPont launched EverpreX™ herbicide for soybean growers to manage resistance
UPL Adds New Herbicide Products for the US Market
Syngenta launches herbicide Lumax for corn in Spain
S-metolachlor Mesotrione Terbuthylazine
Syngenta scientists helps combat difficult weeds through new herbicide Acuron
Atrazine Bicyclopyrone Mesotrione S-metolachlor
China approved Shangyu Nutrichem’s herbicide S-metolachlor
Related CompaniesMore
Hangzhou Qingfeng Imp.& Exp. Co.,Ltd.
Country: China
S-metolachlor Fomesafen Flumioxazin Clethodim Imazapyr Tebuthiuron Azoxystrobin Tebuconazole Oxyfluorfen Metolachlor
Country: China
Picloram Glyphosate 2,4-D Paraquat Lufenuron Chlorpyrifos Chlorothalonil Imidacloprid Cymoxanil Metalaxyl+Cuprous Oxide
Jiangsu Good Harvest-Weien Agrochemical Co., Ltd.
Country: China
Glyphosate S-metolachlor Metaldehyde Metamitron Penoxsulam Florasulam Mesotrione Amicarbazone Pyraclostrobin Mesosulfuron-methyl
Country: China
Glyphosate Fluazinam Clethodim Haloxyfop-P-methyl+Clethodim Bifenthrin Lambda-cyhalothrin Flumorph+Fosetyl-aluminium Flumorph+Mancozeb
Shenzhen Baocheng Chemical Industry CO.,LTD.
Country: China
Glyphosate Emamectin benzoate Lambda-cyhalothrin Pyraclostrobin Bromacil Glufosinate-ammonium Tembotrione Prothioconazole Topramezone Chlorantraniliprole

 0
0 Subscribe
Subscribe