-
Common NameTriazamate
-
中文通用名唑蚜威
-
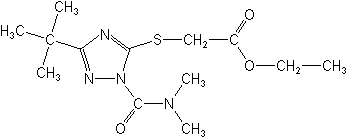
IUPACethyl (3-tert-butyl-1-dimethylcarbamoyl-1H-1,2,4-triazol-5-ylthio)acetate
-
CASethyl [[1-[(dimethylamino)carbonyl]-3-(1,1-dimethylethyl)-1H-1,2,4-triazol-5-yl]thio]acetate
-
CAS No.112143-82-5
-
Molecular FormulaC13H22N4O3S
-
Molecular Structure
-
Category
-
ActivityInsecticide
Triazamate is a plant-systemic aphicide which moves both acropetally and basipetally. It acts primarily as a stomach poison and also has contact activity. The product has a rapid knockdown effect. Reductions in the number of aphids can be seen within one hour of application, with complete control within 24 hours. It also has residual activity lasting 2-3 weeks depending on species. Triazamate is specific to aphids. It is safe to beneficial insects and mites. The company notes that the use of adjuvants improves rainfastness and allows lower application rates to be used.
In field trials, triazamate gave similar or better results than standard insecticides at lower application rates. It is effective on strains of Myzus persicae (peach-potato aphid), Aphis gossypii (cotton aphid) and Phorodon humuli (damson-hop aphid) resistant to certain other insecticides. -
CropUseCropUses:
canola, oilseed rape, sugar beets, fruits, vegetables, hops, potatoes, sunflowers, ornamentals
Oilseed rape
70 g ai/ha
Potatoes
105-140 g ai/ha
Sugar beet
70 g ai/ha
Hops
7-14 g ai/ha
Apples
70 g ai/ha
Sunflowers
70 g ai/ha
Citrus
5-28 g ai/ha
Ornamentals
70-140 g ai/ha
Vegetables
35-140 g ai/ha
-
PremixEmulsion, oil in water, wettable powder.
Type
AI concn
Oil-in-water emulsion (EW)
14% (w/v)
-
Physical PropertiesMolecular weight:314.4; Physical form:White to light tan, crystalline solid, with a slight sulfur odour ( tech.). Density:1.222 (20.5 °C); Melting point:54 °C; Flash point:189 °C (EEC A9); Vapour pressure:0.16 mPa (25 °C); Henry constant:1.26 × 10-4 Pa m3 mol-1 (25 °C, calc.); Partition coefficient(n-octanol and water):logP = 2.15 ( pH 7, 25 °C); pKa:Non-ionising pH 4-9; Solubility:In water 399 ppm ( pH 7, 25 °C). Soluble in dichloromethane and ethyl acetate (tech.).; Stability:Stable under normal storage conditions and at pH ×7.0; DT50 220 d ( pH 5), 49 h ( pH 7), 1 h ( pH 9).;
-
ToxicologyOral:Acute oral LD50 for male rats 100-200, female rats 50-100, mice 54 mg tech./kg. Percutaneous:Acute percutaneous LD50 for rats >5000 mg/kg. Practically non-irritating to the skin; moderate eye irritant (rabbits). Not a skin sensitiser (guinea pigs). Inhalation: LC50 for rats 0.47 mg/l air. ADI:0.012 mg/kg b.w.
-
Environmental ProfileEcotoxicology:
Algae: EC50 (120 h) for Selenastrum capricornutum 27 mg/l; NOEC (120 h) 9.7 mg/l.Bees:Non-toxic; LD50 (96 h, oral) 41 µg/bee; (96 h, contact, for tech.) 27 µg/bee.Birds:Acute oral LD50 (single dose) for bobwhite quail 8 mg/kg. Dietary LC50 (8 d) for mallard ducks 292, quail 411 ppm.Daphnia: LC50 (48 h) 0.014 mg/l.Fish: LC50 (96 h) for bluegill sunfish 0.74, rainbow trout 0.53, sheepshead minnow 5.9 mg/l.Worms: LC50 (14 d) for earthworms 340 mg/kg; NOEC <95 mg/kg.Other aquatic spp.: LC50 (120 h) for mysid shrimp (Mysidopsis bahia) 190 µg/l.Other beneficial spp.:Poecilus cupreus and Aleochara bilineata: no toxic effects at 140 g/ha (lab.). Coccinella septempunctata: 87% effects at 140 g/ha (lab.); no toxic effects in semi-field tests.
Environmental fate:
Very rapidly metabolised by enzyme-catalysed hydrolysis and oxidation in all biological systems studied. Transient metabolites are either degraded further (soils and plants), or are eliminated (vertebrates). Animals:Hydrolysis, followed by decarbamoylation. Soil:DT50 2-6 h. Koc 150-369 (5 soils, 25 °C). Aqueous photolysis DT50 301 d ( pH c. 4). Metabolites found in leachates are not acutely toxic to DapPlant:Hydrolysis, followed by decarbamoylation.
Bobwhite quail
LD50 8 mg/kg
Rainbow trout [96 hrs]
LC50 0.53 mg/litre
Bluegill sunfish [96 hrs]
LC50 0.88 mg/litre
Bee [contact]
LD50 >160μg/bee
Fate in soil:
Triazamate is moderately mobile in soil. It has a half-life of less than one day.Fate in aquatic systems:
The half-life of triazamate in aqueous solution varies from 1 hour to 10 days with decreasing pH. -
Transport InformationHazard Class:O (Obsolete as pesticide, not classified)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe