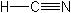-
Common NameHydrogen cyanide
-
中文通用名氰化氢
-
IUPAChydrogen cyanide
-
CAShydrocyanic acid
-
CAS No.74-90-8
-
Molecular FormulaCHN
-
Molecular Structure
-
Category
-
ActivityInsecticide/Rodenticide
-
Physical PropertiesMolecular weight:27.0g/mol; Physical form:Colourless, flammable liquid, with an odour of almonds. Density:0.699 (20℃); Melting point:-15℃; Vapour pressure:961 kPa (25℃); Henry constant:2.59 Pa m3mol-1 (calc.); pKa:Weak acid; Solubility:Completely miscible with water. Soluble in ethanol and diethyl ether.; Stability:Stable, in the pure form. In the liquid form and in aqueous solution, stabilised by the addition of small quantities of mineral acids.
-
ToxicologyOral Acute oral LD50 for rats 10-15 mg/kg.Skin and eye Rapidly absorbed by skin. Eye irritant.Inhalation Exposure for 30 min at 0.36 mg/l air is fatal to man.ADI (JMPR) 0.05 mg/kg b.w. [1965].
-
Environmental ProfileEcotoxicology:
Bees:Toxic to bees, but not hazardous if used as directed.Birds:Toxic to birds.Fish:Toxic to fish.
Environmental fate:
Animals:The cyanide ion is metabolised mainly to thiocyanate. -
Transport InformationHazard Class:O (Obsolete as pesticide, not classified)
Porduct NewsMore
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos
ADAMA Canada moves forward with lambda-cyhalothrin sales for 2023
Mexico: Import of glyphosate drops from 16,500 tons to 8,200 tons
Glyphosate prices drop 30% in Argentina
One year extension of EU glyphosate approval failed to pass in the latest meeting

 0
0 Subscribe
Subscribe