-
Common NameOxabetrinil
-
中文通用名解草腈
-
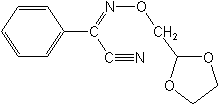
IUPAC(Z)-1,3-dioxolan-2-ylmethoxyimino(phenyl)acetonitrile
-
CAS(αZ)-α-[(1,3-dioxolan-2-yl)methoxyimino]benzeneacetonitrile
-
CAS No.94593-79-0
-
Molecular FormulaC12H12N2O3
-
Molecular Structure
-
Category
-
ActivityHerbicide safener
-
Physical PropertiesMolecular weight:232.2; Physical form:Colourless crystals. Density:1.33 (20℃); Composition:Material comprises (Z)- and (E)- isomers. Melting point:77.7 ℃; Vapour pressure:0.53 mPa (20℃); Henry constant:6.15 × 10-3 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 2.76 (rp-tlc); Solubility:In water 20 mg/l (20 ℃). In acetone 250, cyclohexanone 300, toluene 220, methanol 30, hexane 5.6, n-octanol 12, xylene 150, dichloromethane 450 (all in g/ kg, 20℃).; Stability:Stable up to 240 ℃. No significant hydrolysis at pH 5-9 within 30 d.
-
ToxicologyOral:Acute oral LD50 for rats >5000 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats >5000 mg/kg. Minimal skin and eye irritation (rabbits). Not a skin sensitiser. Inhalation: LC50 (4 h) for rats >1.45 mg/l air. ADI:0.12 mg/ kg.
-
Environmental ProfileEcotoxicology:
Algae: EC50 (96 h) for Selenastrum capricornutum 10.7 mg/l.Bees: LD50 (oral, 24 h) >20 μg/bee; (contact) >1000 ppm.Birds: LD50 for Japanese quail >2500 mg/ kg; (8 d) for bobwhite quail >5000, Pekin ducks >1000 mg/ kg.Daphnia: LC50 (48 h) 8.5 mg/l.Fish: LC50 (96 h) for trout 7.1, bluegill sunfish 12 mg/l.
Environmental fate:
Animals:In rats, following ingestion, oxabetrinil is rapidly and fully excreted, predominantly via urine.Plant:Opening of the dioxolane ring, hydroxylation, glycosylation to water-soluble conjugation products. -
Transport InformationHazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe