-
Common NamePebulate
-
中文通用名克草敌
-
IUPACS-propyl butyl(ethyl)thiocarbamate
-
CASS-propyl butylethylcarbamothioate
-
CAS No.1114-71-2
-
Molecular FormulaC10H21NOS
-
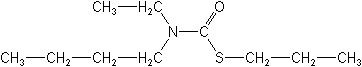
Molecular Structure
-
Physical PropertiesMolecular weight:203.3; Physical form:Colourless or yellow liquid, with an aromatic odour. Density:0.956 (20 °C); Composition:Tech. material is 95% pure. Flash point:124 °C; Vapour pressure:9 Pa (30 °C); 4.7 Pa (25 °C); Henry constant: 20 Pa m3 mol-1 (20 °C, calc.); Partition coefficient(n-octanol and water):logP = 3.83; Solubility:In water 60 mg/l (20 °C). Miscible with most organic solvents, e.g. acetone, benzene, toluene, xylene, methanol, isopropanol, kerosene.; Stability:Stable up to 200 °C. In water, DT50 (40 °C) 11 d (pH 4 and pH 10), 12 d (pH 7).
-
ToxicologyOral:Acute oral LD50 for rats 1120, mice 1652 mg/ kg. Percutaneous:Acute percutaneous LD50 for rabbits 4640 mg/kg. Slight irritation of skin, eyes and mucous membranes (rabbits). Not a skin sensitiser (guinea pigs). Inhalation: LC50 (4 h) for female rats >3.5 mg/l. ADI:( JMPR) 0.003 mg/ kg b.w. [1995].
-
Environmental Profile.Ecotoxicology:
Bees:Non-toxic to bees at 0.011 mg/bee.Birds:Dietary LC50 (7 d) for bobwhite quail 8400 mg/ kg diet.Daphnia: LC50 (48 h) 5.9 mg/l.Fish: LC50 (96 h) for rainbow trout and bluegill sunfish c. 7.4 mg/l; (48 h) for silver mullet 6.25, killifish 7.78 mg/l.
Environmental fate:
Animals:In animals, pebulate is rapidly metabolised. In rats, following oral administration, c. 50% was expired as CO2, 25% excreted in the urine, and 5% excreted in the faeces, within 3 days.Soil:In soil, pebulate disappears mainly by microbial degradation to the mercaptan, ethylbutylamine, and CO2. DT50 in soil 2-3 w.Plant:In plants, pebulate is rapidly metabolised to CO2 and naturally-occurring plant constituents. WATER SOLUBILITY: Tech 60 ppm at 58°F -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos

 0
0 Subscribe
Subscribe