-
Common NamePhorate
-
中文通用名甲拌磷
-
IUPACO,O-diethyl S-ethylthiomethyl phosphorodithioate
-
CASO,O-diethyl S-[(ethylthio)methyl] phosphorodithioate
-
CAS No.298-02-2
-
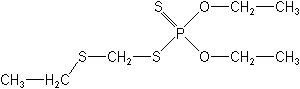
Molecular FormulaC7H17O2PS3
-
Molecular Structure
-
Category
-
ActivityInsecticide, acaricide, nematicide
-
Physical PropertiesMolecular weight:260.4; Physical form:Colourless liquid ( tech.). Density:1.167 ( tech., 25 °C); Composition:Tech. phorate is >90% pure. Melting point:<-15 °C ( tech.); Flash point:>110 °C (Setaflash closed cup); Vapour pressure:85 mPa (25 °C); Henry constant:5.9 × 10-1 Pa m3 mol-1 (calc.); Partition coefficient(n-octanol and water):logP = 3.92; Solubility:In water 50 mg/l (25 °C). Miscible with alcohols, ketones, ethers, esters, aromatic, aliphatic and chlorinated hydrocarbons, dioxane, vegetable oils, and other organic solvents.; Stability:Stable under normal storage conditions for at least 2 years. Aqueous solutions degraded by light ( DT50 1.1 d); stability to hydrolysis optimum at pH 5-7, DT50 3.2 d.
-
ToxicologyOral:Acute oral LD50 for male rats 3.7, female rats 1.6, mice c. 6 mg/kg. Percutaneous:Acute percutaneous LD50 for male rats 6.2, female rats 2.5, guinea pigs 20-30, male rabbits 5.6, female rabbits 2.9 mg/kg. Values for GR depend on a.i. content, carrier, test method and animal sp Inhalation: LC50 (1 h) for male rats 0.06, female rats 0.011 mg/l. Phytotoxicity:Phytotoxic to apples and tobacco. Phytotoxicity is possible in beet, carrots, beans, maize, an
-
Environmental ProfileEcotoxicology:
Bees:Toxic to bees; LD50 (topical) 10µg/bee.Birds:Acute oral LD50 for mallard ducks 0.62, ring-necked pheasants 7.1 mg/ kg.Fish: LC50 (96 h) for rainbow trout 0.013, channel catfish 0.28 mg/l.
Environmental fate:
Animals:In animals, phorate is metabolically oxidised to the sulfoxide and sulfone, and their phosphorothioate analogues, followed by hydrolysis to dithio-, thio-, and orthophosphoric acids.Soil:In soil, metabolic oxidation gives the sulfoxide and sulfone, and their phosphorothioate analogues, and these then undergo hydrolysis, although the sulfone can persist under certain conditions (D. L. Suett, Pestic. Sci., 1975, 6,Plant:Degradation is similar to that in animals. WATER SOLUBILITY: 50 mg/l at 25°C . -
Transport InformationHazard Class:Ia(Extremely hazardous)
Porduct NewsMore
Phorate and acetochlor recommended to be listed in Annex III of the Rotterdam Convention
Brazil Anvisa approves to ban two ais
Related CompaniesMore
Country: India
Country: India
HPM Chemicals & Fertilizers Ltd.
Country: India

 0
0 Subscribe
Subscribe