-
Common NameProthiofos
-
中文通用名丙硫磷
-
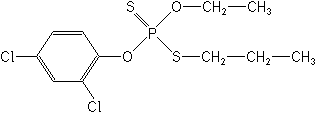
IUPAC(RS)-(O-2,4-dichlorophenyl O-ethyl S-propyl phosphorodithioate)
-
CASO-(2,4-dichlorophenyl) O-ethyl S-propyl phosphorodithioate
-
CAS No.34643-46-4
-
Molecular FormulaC11H15Cl2O2PS2
-
Molecular Structure
-
Category
-
ActivityInsecticide
-
Physical PropertiesMolecular weight:345.2; Physical form:Colourless liquid, with a weak characteristic odour. Density:1.31 (20 °C); Flash point:>110 °C; Vapour pressure:0.3 mPa (20 °C); 0.6 mPa (25 °C); Partition coefficient(n-octanol and water):logP = 5.67 (20 °C); Solubility:In water 0.07 mg/l (20 °C). In dichloromethane, isopropanol, toluene >200 g/l (20 °C).; Stability:Hydrolysis DT50 in buffer (22 °C) 120 d ( pH 4), 280 d ( pH 7), 12 d ( pH 9). Photodegradation DT50 13 h.
-
ToxicologyOral:Acute oral LD50 for male rats 1569, female rats 1390, mice c. 2200 mg/ kg. Percutaneous:Acute percutaneous LD50 (24 h) for rats >5000 mg/kg. Not irritating to skin and eyes (rabbits). Skin sensitiser. Inhalation: LC50 (4 h) for rats >2.7 mg/l air (aerosol). Phytotoxicity:Russetting is possible with Golden Delicious apples. ADI:0.019 mg/ kg b.w.
-
Environmental ProfileEcotoxicology:
Algae: ErC50 for Scenedesmus subspicatus 2.3 mg/l.Bees:Not hazardous to bees if used as recommended.Birds:Acute oral LD50 for Japanese quail 100-200 mg/ kg.Daphnia: LC50 (48 h) 0.014 mg/l.Fish: LC50 (96 h) for golden orfe 4-8, rainbow trout 0.5-1 mg/l (500 g/l EC formulation).
Environmental fate:
Animals:In rats, prothiofos is rapidly absorbed and metabolised. At 72 h after administration, 98% of the dose was excreted. Metabolic routes include oxidation to prothiofos-oxon, hydrolysis to 2,4-dichlorophenol which is further conjugated, and cleavage of the pSoil:Prothiofos is very strongly adsorbed in soil. DT50 under field conditions is 1-2 mo. In soil, dechlorination to 4-chloroprothiofos and oxidation to prothiofos-oxon occurs, as well as hydrolysis to 2,4-dichlorophenol which is finPlant:In plants, prothiofos is hydrolysed to 2,4-dichlorophenol which is further conjugated. Formation of prothiofos-oxon and cleavage of the propyl group also occurs. WATER SOLUBILITY: 0.07 mg/l at 20° C -
Transport InformationSignal Word:CAUTION; Hazard Class:II(Moderately hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe