-
Common NameSimazine
-
中文通用名西玛津
-
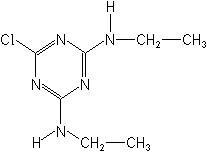
IUPAC6-chloro-N2,N4-diethyl-1,3,5-triazine-2,4-diamine
-
CAS6-chloro-N,N′-diethyl-1,3,5-triazine-2,4-diamine
-
CAS No.122-34-9
-
Molecular FormulaC7H12ClN5
-
Molecular Structure
-
Category
-
ActivityHerbicide
-
PremixWettable powder, water dispersible granule, liquid, and granular. Premix Parters: cacodylic acid; cacodylic acid MSMA;
-
Physical PropertiesMolecular weight:201.7; Physical form:Colourless powder. Density:1.33 (22 °C); Composition:Tech. grade is ×97% pure. Melting point:225-227 °C (decomp.); Vapour pressure:2.94 × 10-3 mPa (25 °C) ( OECD 104); Henry constant:5.6 × 10-5 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 2.1 (25 °C, unionised); pKa:1.62 (20 °C), v. weak base; Solubility:In water 6.2 mg/l ( pH 7, 20 °C). In ethanol 570, acetone 1500, toluene 130, n-octanol 390, n-hexane 3.1 (all in mg/l, 25 °C).; Stability:Relatively stable in neutral, weakly acidic and weakly alkaline media. Rapidly hydrolysed by stronger acids and bases; DT50 ( calc.) 8.8 d ( pH 1), 96 d ( pH 5), 3.7 d ( pH;
-
ToxicologyOral:Acute oral LD50 for rats 500-10 000, Chinese hamsters >5000 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >2000 mg/kg. Non-irritating to skin and eyes (rabbits). Inhalation: LC50 (4 h) for rats >5.5 mg/l. Phytotoxicity:Phytotoxic to a number of crops, including sugar beet, tobacco, tomatoes, cucurbits, clover, rice, soya beans, lettuce, oats, and many vegetables (e.g. spinach, onions, carrots, crucifers, etc.).
-
Environmental ProfileEcotoxicology:
Algae: EC50 (72 h) for Scenedesmus subspicatus 0.042 mg/l; (5 d) for Selenastrum capricornutum 0.26 mg/l.Bees:LD50 (48 h, oral and topical) >99mg/bee.Birds:Acute oral LD50 for mallard ducks >2000 mg/kg. Dietary LC50 (8 d) for mallard ducks 10 000, Japanese quail >5000 mg/kg.Daphnia:LC50 (48 h) >100 mg/l; (21 d) 0.29 mg/l.Fish: LC50 (96 h) for bluegill sunfish 90, rainbow trout >100, crucian carp >100, guppies 49 mg/l.Worms: LC50 (14 d) for earthworms >1000 mg/kg.
Environmental fate:
Animals:In mammals, following oral administration, 65-97% is eliminated within 24 h as the de-ethylated metabolite (Y. Deng et al., J. Agric. Food Chem., 1990, 38, 1411). Elimination of low rates is primarily in the urinSoil:Major metabolites under all conditions are desethylsimazine and hydroxysimazine. Microbial breakdown in soil results in degradation of simazine at very variable rates; DT50 27-102 d (median 49 d); temperature and soil moisture aPlant:Readily metabolised by tolerant plants to the herbicidally-inactive 6-hydroxy analogue and amino acid conjugates. The hydroxysimazine is further degraded by dealkylation of the side-chains and by hydrolysis of the resulting amino groups on the ring, with -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Sipcam's Coastal Herbicide registered in US
Related CompaniesMore
Lianyungang Liben Crop Science Co.,Ltd.
Country: China
Terbuthylazine Paraquat Atrazine Ametryn Prometryn Pyraclostrobin Isoprothiolane Simazine Azoxystrobin Chlorpyrifos Nitenpyram
Jiangsu Good Harvest-Weien Agrochemical Co., Ltd.
Country: China
Glyphosate S-metolachlor Metaldehyde Metamitron Penoxsulam Florasulam Mesotrione Amicarbazone Pyraclostrobin Mesosulfuron-methyl
Country: China
Glufosinate-ammonium 2,4-D MCPA Dicamba Propanil Clethodim Glyphosate Captan Flumioxazin Sulfentrazone
Country: China
Clethodim Hexazinone Mesotrione Abamectin Acetamiprid Dimethoate Fipronil Imidacloprid Fosetyl-aluminium 2,4-D
Shandong Binnong Technology Co., Ltd.
Country: China
Atrazine Ametryn Metolachlor S-metolachlor Terbuthylazine Fomesafen Mesosulfuron-methyl Glufosinate-ammonium Bentazone Pendimethalin

 0
0 Subscribe
Subscribe