-
Common NameTebuthiuron
-
中文通用名丁噻隆
-
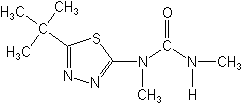
IUPAC1-(5-tert-butyl-1,3,4-thiadiazol-2-yl)-1,3-dimethylurea
-
CASN-[5-(1,1-dimethylethyl)-1,3,4-thiadiazol-2-yl]-N,N′-dimethylurea
-
CAS No.34014-18-1
-
Molecular FormulaC9H16N4OS
-
Molecular Structure
-
Physical PropertiesMolecular weight:228.3; Physical form:Colourless, odourless solid. Melting point:161.5-164 °C (decomp.); Vapour pressure:0.27 mPa (25 °C); Henry constant:2.47 × 10-5 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 1.79 ( pH 7, 25 °C); Solubility:In water 2.5 g/l (25 °C). In benzene 3.7, hexane 6.1, 2-methoxyethanol 60, acetonitrile 60, acetone 70, methanol 170, chloroform 250 (all in g/l, 25 °C).; Stability:Stable at 52 °C (highest storage temperature tested). Stable in aqueous media between pH 5 and 9. On hydrolysis, DT50 (25 °C) >64 d ( pH 3, 6 and 9).;
-
ToxicologyOral:Acute oral LD50 for mice 579, rats 644, rabbits 286, dogs >500, cats >200 mg/kg. Percutaneous:No skin irritation in rabbits when applied at 200 mg/kg; no eye irritation when applied at 71mg/kg. Phytotoxicity:Not to be applied near desirable trees or plants. ADI:0.15 mg/kg b.w.
-
Environmental ProfileEcotoxicology:
Algae: EC50 for Anabaena 4.06, Navicula 0.081, Selenastrum 0.05, Skeletonema 0.05 ppm.Bees:LD50 >100 µg/bee.Birds:Acute oral LD50 for chickens, bobwhite quail, and mallard ducks >500 mg/kg. In 1 mo feeding trials, chickens receiving 1000 mg/kg showed no ill-effects.Daphnia: LC50 297 ppm.Fish: LC50 (96 h) for rainbow trout 144, goldfish and fathead minnow >160, bluegill sunfish 112 mg/l.Other aquatic spp.: EC50 for Lemna 0.135 ppm.
Environmental fate:
Animals:The major metabolites in mammals were formed by N-demethylation of the substituted urea side-chain (D. M. Morton & D. G. Hoffman, J. Toxicol. Environ. Health, 1976, 1, 757-768).Soil:Some microbial breakdown occurs in soil, but this is not the predominant mode of degradation. Loss due to photodecomposition and volatilisation is negligible. Half-life in soil is considerably greater in soils with low moisture content, and in high organiPlant:In plants, the principal metabolic pathways involve N-demethylation and hydroxylation of the tert-butyl side-chain. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Nutrichem To Purchase Dow AgroSciences’ Tebuthiuron Herbicide Business
Related CompaniesMore
Hangzhou Qingfeng Imp.& Exp. Co.,Ltd.
Country: China
S-metolachlor Fomesafen Flumioxazin Clethodim Imazapyr Tebuthiuron Azoxystrobin Tebuconazole Oxyfluorfen Metolachlor
Country: China
Oxyfluorfen Tebuconazole Metolachlor Azoxystrobin Clopyralid Triclopyr Tebuthiuron Acifluorfen Clethodim Imazapyr Picloram Pretilachlor Bifenthrin Amitraz Mesotrione
Country: China
Fluroxypyr-meptyl Clodinafop-propargyl Glufosinate-ammonium Picloram Clopyralid Triclopyr-butotyl Flumioxazin Triclopyr Diquat Epoxiconazole
Yifan Biotechnology Group Co., Ltd.
Country: China
Fipronil Difenoconazole Metalaxyl Picoxystrobin Oxyfluorfen Clethodim Clodinafop-propargyl Metalaxyl-M Flumioxazin Prothioconazole
Country: China
Carbofuran Cartap Carbosulfan Abamectin Emamectin benzoate Bifenthrin Glufosinate ammonium Pyraclostrobin Difenoconazole Boscalid

 0
0 Subscribe
Subscribe