-
Common NameTerbacil
-
中文通用名特草定
-
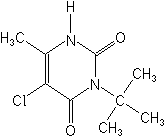
IUPAC3-tert-butyl-5-chloro-6-methyluracil
-
CAS5-chloro-3-(1,1-dimethylethyl)-6-methyl-2,4(1H,3H)-pyrimidinedione
-
CAS No.5902-51-2
-
Molecular FormulaC9H13ClN2O2
-
Molecular Structure
-
Physical PropertiesMolecular weight:216.7; Physical form:Colourless crystals. Density:1.34 (25 °C); Composition:Tech. is 97%. Melting point:175-177 °C; Vapour pressure:0.0625 mPa (29.5 °C); Henry constant:1.3 × 10-5 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 1.91; pKa:9.5; Solubility:In water 710 mg/l (25 °C). In dimethylformamide 337, cyclohexanone 220, methyl isobutyl ketone 121, butyl acetate 88, xylene 65 (all in g/kg, 25 °C). Sparingly soluble in mineral oils and aliphatic hydrocarbons. Readily s; Stability:Very stable, even at m.p. Stable in aqueous alkaline media at room temperature. Stable 31 d in the dark in aqueous solution (c. pH 6, 25 °C), and in an aqueous solution containing 0.05 M ferric chloride (c
-
ToxicologyOral:Acute oral LD50 for rats 934 mg/kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/kg. Mild eye irritant; slight skin irritant. Not a skin sensitiser. Inhalation: LC50 (4 h) for rats >4.4 mg/l.
-
Environmental ProfileEcotoxicology:
Bees:Non-toxic to bees.Birds:Dietary LC50 (8 d) for Pekin ducklings >56 000, pheasant chicks >31 450 mg/kg diet.Daphnia: LC50 (48 h) 68 ppm.Fish: LC50 (96 h) for rainbow trout 46.2 mg/l.Other aquatic spp.: LC50 (48 h) for fiddler crab >1000 mg/l.
Environmental fate:
Animals:The principal biotransformation pathways are hydroxylation of the 6-methyl group, and replacement of the 5-chloro group with a hydroxy group (B. C. Mayo et al., Proc. Br. Crop Prot. Conf. - Pests Dis., 1988, 2, 6Soil:Undergoes microbial decomposition in moist soil. In top soil, 50% still remains 5-7 months after applying 4.5 kg/ha (P. B. Marriage, Weed Res., 1977, 17, 219-225).Plant:In alfalfa, 12% of terbacil plus its metabolites are still found 6-8 months after application (R. F. Holt & H. L. Pease, J. Agric. Food Chem., 1977, 25, 373). -
Transport InformationHazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Related CompaniesMore
Jiangsu Luye Agrochemicals Co., Ltd.
Country: China
Azoxystrobin Lenacil Metribuzin Propyzamide Asulam Prochloraz Terbacil Indoxacarb Thiamethoxam Validamycin Imidacloprid Bromoxynil octanoate

 0
0 Subscribe
Subscribe