-
Common NameTerbutryn
-
中文通用名特丁净
-
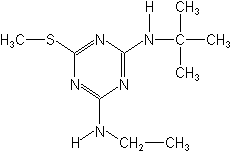
IUPACN2-tert-butyl-N4-ethyl-6-methylthio-1,3,5-triazine-2,4-diamine
-
CASN-(1,1-dimethylethyl)-N′-ethyl-6-(methylthio)-1,3,5-triazine-2,4-diamine
-
CAS No.886-50-0
-
Molecular FormulaC10H19N5S
-
Molecular Structure
-
Category
-
ActivityHerbicide
-
PremixMetolachlor+Terbutryn
Wettable powder, flowable, liquid. Premix Parters: carbendazim; chlorothalonil; prochloraz; sulfur;
-
Physical PropertiesMolecular weight:241.4; Physical form:White powder. Density:1.12 (20 °C); Composition:Tech. is 96%. Melting point:104-105 °C; Vapour pressure:0.225 mPa (25 °C) ( OECD 104); Henry constant:1.5 × 10-3 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 3.65 (25 °C, unionised); pKa:4.3, weak base; Solubility:In water 22 mg/l (22 °C). In acetone 220, hexane 9, n-octanol 130, methanol 220, toluene 45 (all in g/l, 20 °C). Also readily soluble in dioxane, diethyl ether, xylene, chloroform, carbon tetrachloride, dimethylformamide. Slightl; Stability:Stable under normal conditions. The methylthio group is hydrolysed in the presence of strong acids or alkalis. At 70 °C, no significant hydrolysis occurs at pH 5, 7, or 9.;
-
ToxicologyOral:Acute oral LD50 for rats 2500, mice 500 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >2000, rabbits >20 000 mg/kg. Not a skin or eye irritant (rabbits). Not a skin sensitiser (guinea pigs). Inhalation: LC50 (4 h) for rats >2200 mg/m3 air. Phytotoxicity:Not safe for post-emergence use in cereals which are under stress. ADI:0.0022 mg/kg.
-
Environmental ProfileEcotoxicology:
Algae: EC50 (7 d) for Selenastrum capricornutum 0.013 mg/l.Bees: Not toxic to bees. LD50 (oral) >225 µg/bee; (contact) >100 µg/bee.Birds:Dietary LC50 (5 d) for bobwhite quail 5000, mallard ducks >4640 mg/kg b.w.Daphnia: LC50 (48 h) 2.66 mg/l.Fish:LC50 (96 h) for rainbow trout 1.1, bluegill sunfish 1.3, carp 1.4.Worms: LC50 for Eisenia foetida 170 mg/kg.Other aquatic spp.: EC50 (48 h) for Quahog clam 5.6 mg/l.
Environmental fate:
Animals:In mammals, following oral administration, 73-85% is eliminated as the dealkylated hydroxy metabolite in the faeces within 24 hours (Y. Deng et al., J. Agric. Food Chem., 1990, 38, 1411).Soil:Soil micro-organisms play an important role in the degradation of terbutryn. Residual activity in soil is 3-10 weeks, depending upon rate of application, soil type, and weather. DT50 in soil 14-50 d. Koc Plant:In plants, terbutryn is degraded in a manner similar to other methylthio-s-triazines, viz. by oxidation of the methylthio group to hydroxy metabolites, and by dealkylation of the side-chains. Conjugates are also formed. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Related CompaniesMore
Lianyungang Liben Crop Science Co.,Ltd.
Country: China
Terbuthylazine Paraquat Atrazine Ametryn Prometryn Pyraclostrobin Isoprothiolane Simazine Azoxystrobin Chlorpyrifos Nitenpyram
Country: China
Glufosinate-ammonium 2,4-D MCPA Dicamba Propanil Clethodim Glyphosate Captan Flumioxazin Sulfentrazone
Shandong Binnong Technology Co., Ltd.
Country: China
Atrazine Ametryn Metolachlor S-metolachlor Terbuthylazine Fomesafen Mesosulfuron-methyl Glufosinate-ammonium Bentazone Pendimethalin
Zhejiang Zhongshan Chemical Industry Group Co., Ltd.
Country: China
Ametryn Terbuthylazine Metamitron S-metolachlor Picoxystrobin Mesotrione Bentazone Atrazine Isoxaflutole Florasulam Metolachlor

 0
0 Subscribe
Subscribe