-
Common NameButoxycarboxim
-
中文通用名丁酮砜威
-
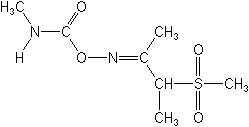
IUPAC(EZ)-3-mesylbutanone O-methylcarbamoyloxime
-
CAS3-(methylsulfonyl)-2-butanone O-[(methylamino)carbonyl]oxime
-
CAS No.34681-23-7
-
Molecular FormulaC7H14N2O4S
-
Molecular Structure
-
Category
-
ActivityInsecticide, acaricide
-
Physical PropertiesMolecular weight:222.3; Physical form:Colourless crystals. Density:1.21 g/cm3 (20 °C); Composition:Tech. butoxycarboxim contains the (E)- and (Z)- isomers in a ratio of 85-90:15-10. Melting point:85-89 °C (tech.); Vapour pressure:0.266 mPa (20 °C) ( tech.); Henry constant:2.83 × 10-7 Pa m3 mol-1 ( calc.); Solubility:In water 209 g/l (20 °C). Readily soluble in polar organic solvents; slightly soluble in non-polar solvents. In chloroform 186, acetone 172, isopropanol 101, toluene 29, carbon tetrachloride 5.3, cyclohexane 0.9, heptane 0.1 (all in g/l, 20 °C).; Stability:Aqueous hydrolysis DT50, 501 d ( pH 5), 18 d ( pH 7), 16 d ( pH 9). Stable to u.v. light. Thermally stable up to 100 °C.;
-
ToxicologyOral:Acute oral LD50 for rats 458, rabbits 275 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >2000 mg/kg. ADI:( JMPR) No ADI [1985]; (Bundes Gesundheitsamt, Berlin) 0.02 mg/kg.
-
Environmental ProfileEcotoxicology:
Bees:Not hazardous to bees (special test with 'Plant Pin').Birds:Acute oral LD50 for hens 367 mg/kg.Daphnia: LC50 (96 h) 500mg/l.Fish: LC50 (96 h) for carp 1750, rainbow trout 170 mg/l.
Environmental fate:
Animals:In mammals, following oral administration, eliminated in the urine both in the unchanged form and as metabolites. There is no accumulation in tissue.Soil: DT50 in soil 41-44 d (20 °C). Butoxycarboxim diffuses from the pasteboard stick into the soil within 2 days, activity commences after c. 3 days, reaches its full extent in 7-14 days, and remains for 4-8 weeks, until the -
Transport InformationSignal Word:WARNING; Hazard Class:II(Moderately hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe