-
Common NameHalosulfuron-methyl
-
中文通用名氯吡嘧磺隆
-
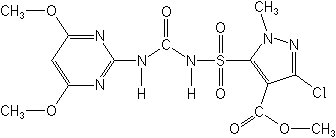
IUPACmethyl 3-chloro-5-(4,6-dimethoxypyrimidin-2-ylcarbamoylsulfamoyl)-1-methylpyrazole-4-carboxylate
-
CASmethyl 3-chloro-5-[[[[(4,6-dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]-1-methyl-1H-pyrazole-4-carboxylate
-
CAS No.100784-20-1
-
Molecular FormulaC13H15ClN6O7S
-
Molecular Structure
-
Category
-
ActivityHerbicide
Halosulfuron-methyl is a post-emergence herbicide. It is rapidly absorbed via the foliage and the roots and translocated to other parts of the plant where it quickly arrests growth, leading to the death of the plant. The compound provides residual soil activity resulting in season long control. The product does not injure crops planted in rotation. A non-ionic surfactant or crop oil concentrate must be applied with the herbicide.
In rice, Permit is labelled for control of nutsedge (Cyperus spp) plus cocklebur, redroot pigweed and smartweed. The product may be applied by ground or by air, prior to planting through to establishment of the permanent flood in dry-seeded rice. It may be tank-mixed with labelled preplant burndown herbicides to increase control of nutsedge. Due to the low residual effect, two treatments may be required to control weeds emerging after application.
Halosulfuron-methyl exhibits a high degree of crop safety in cereals and turf. In addition to this, the low volatility of the compound means that adjacent crops remain unaffected by it. When tested in sequence with maize insecticides, the product caused less crop damage than established products.
Uptake and translocation studies in purple nutsedge showed that halosulfuron-methyl is rapidly taken up by the leaves with most of the applied material being absorbed within 24 hours. The radiolabel was rapidly translocated to the aerial parts and the roots and rhizomes. Unlike glyphosate, accumulation of halosulfuron-methyl in daughter plants ceased within 24 hours of application.
A greenhouse study conducted in 2005 demonstrated that the addition of halosulfuron to clethodim antagonised the control of giant foxtail by 15% when applied as a crop oil concentrate. Whilst tank mixing halosulfuron with sethoxydim antagonised control of giant foxtail when applied with non-ionic surfactant and smooth crabgrass when applied with crop oil concentrate. -
CropUseCropUses:
almonds, asparagus, aubergines, beechnuts, Brazil nuts, butternuts, cantaloupes, cashews, chestnuts, chinquapins, cotton, cucumbers, dry beans, filberts, hickory nuts, lima beans, macadamia nuts, maize, melons, pasture, pecans, peppers, pistachios, pumpkins, rice, snap beans, sorghum, sugar canes, tomatoes, turf, walnuts, winter squash, vegetables
Maize
36-70 g ai/ha (post-emergence), 73-105 g ai/ha (pre-emergence)
Turf
35-140 g ai/ha, 200-400 g ai/ha (Japan)
-
PremixNicosulfuron+Halosulfuron-methyl
Nicosulfuron+Fluroxypyr-meptyl+Halosulfuron-methyl
Mesotrione+Nicosulfuron+Halosulfuron-methyl
Mefenacet+Halosulfuron-methyl+Mesotrione
Halosulfuron-methyl+Mesotrione
Halosulfuron-methyl+Flucarbazone-Na
Florasulam+Halosulfuron-methyl
Ametryn+Halosulfuron-methyl
-
Physical PropertiesMolecular weight:434.8g/mol; Physical form:White powder. Density:1.618 g/ml (25℃); Melting point:175.5-177.2℃; Vapour pressure:<0.01 mPa (25℃); Partition coefficient(n-octanol and water):logP = -0.0186 ( pH 7, 23℃); pKa:3.44 (22℃); Solubility:In water 0.015 ( pH 5), 1.65 ( pH 7) g/l (20℃). In methanol 1.62 g/l (20℃).; Stability:Stable under normal storage conditions.
-
ToxicologyOral:Acute oral LD50 for rats 8866, mice 11 173 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats >2000 mg/kg. Non-irritating to skin; slightly irritating to eyes (rabbits). Non-sensitising to skin (guinea pigs). Inhalation: LC50 (4 h) for rats >6.0 mg/l. Phytotoxicity:Some hybrids or varieties of sweet corn and popcorn may be sensitive.
-
Environmental Profile
Ecotoxicology:
Algae: EC50 (5 d) for green algae (Selenastrum capricornutum) 0.0053, blue-green algae (Anabaena flos-aquae) 0.158 mg/l.Bees: LD50 (dermal) >100 μg/bee.Birds:Acute oral LD50 for bobwhite quail >2250 mg/kg. Dietary LC50 (5 d) for bobwhite quail and mallard ducks >5620 ppm.Daphnia: EC50 (48 h) >107 mg/l.Fish: LC50 (96 h) for bluegill sunfish >118, rainbow trout >131 mg/l.Other aquatic spp.: EC50 (96 h, flow through) for oysters 116 ppm, mysid shrimp (Mysidopsis bahia) 106 mg/l. IC50 (14 d) for Lemna gibba 0.038 μg/l.
Environmental fate:
Animals:Rapidly eliminated from rats, in urine and faeces. The major metabolite is desmethyl halosulfuron-methyl. Further demethylation or hydroxylation of the pyrimidine ring yielded several minor mono- and di- hydroxylated metabolites.Soil:Extensively metabolised in soil. In acidic soils, by hydrolytic cleavage of the sulfonylurea bridge to give aminopyrimidine and 3-chlorosulfonamide ester metabolites, which can undergo further hydrolysis to the acid. Plant:The major metabolite in maize is 3-chloro-1-methyl-5-sulfamoylpyrazole-4-carboxylic acid (cleavage of urea bridge and ester hydrolysis). -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Bayer’s herbicide Tribute Total registered in California
Foramsulfuron Thiencarbazone-methyl Halosulfuron-methyl
Related CompaniesMore
Country: India
Oxyfluorfen Kasugamycin Carfentrazone-ethyl Bispyribac-sodium Pyrithiobac-sodium Quizalofop-P-ethyl Halosulfuron-methyl Carbendazim+mancozeb Tricyclazole Flusilazole Flusilazole+Carbendazim Iprovalicarb+propineb Propineb Difenoconazole+Propiconazole Isoprothiolane Kasugamycin+copper oxychloride Azoxystrobin+difenoconazole Azoxystrobin+Tebuconazole Propargite Cartap hydrochloride Bifenthrin Carbosulfan Emamectin benzoate Acetamiprid Buprofezin Spinosad Chlorantraniliprole Chlorantraniliprole Spiromesifen Imidacloprid Flubendiamide Cyflumetofen Pyriproxyfen Cartap hydrochloride Pendimethalin Pendimethalin Imazethapyr Clodinafop-propargyl Paraquat dichloride Chlorimuron-ethyl 2,4-D dimethyl amine salt Butachlor Copper oxychloride Carbendazim Copper hydroxide Hexaconazole Kasugamycin Carbendazim+mancozeb Carboxin+thiram Carboxin+thiram Propiconazole Imidacloprid Cartap hydrochloride Cartap hydrochloride Sulfoxaflor Fipronil Lambda-cyhalothrin Lambda-cyhalothrin Quinalphos Phenthoate Chlorpyrifos Dinotefuran Fipronil Fipronil Triazophos Monocrotophos Cypermethrin Cypermethrin Chlorantraniliprole Lambda-cyhalothrin Diafenthiuron Chlorpyrifos+cypermethrin

 0
0 Subscribe
Subscribe