-
Common NameChlorfenapyr
-
中文通用名虫螨腈
-
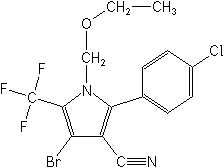
IUPAC4-bromo-2-(4-chlorophenyl)-1-ethoxymethyl-5-trifluoromethyl-1H-pyrrole-3-carbonitrile
-
CAS4-bromo-2-(4-chlorophenyl)-1-(ethoxymethyl)-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile
-
CAS No.122453-73-0
-
Molecular FormulaC15H11BrClF3N2O
-
Molecular Structure
-
Category
-
ActivityAcaricide/Miticide,insecticide.
Chlorfenapyr is a pro-insecticide. On assimilation by the pest, de-alkylation of the pyrrole nucleus occurs revealing the biologically active moiety. The compound is primarily a stomach poison but also has contact activity. Chlorfenapyr has excellent translaminar activity. Depending upon conditions, residual activity may last for more than two weeks. The company reports that there is no cross-resistance to commercially available products and that chlorfenapyr is safe to crops at the recommended rate. Chlorfenapyr is an excellent resistance management tool as a result of its novel mode of action.
Chlorfenapyr has been registered for use against Helicoverpa armigera on cotton in Australia since 1997 where it is part of the Australian Helicoverpa resistance management strategy. During the 1997/8 cotton season it was evident, however, that a low frequency of H armigera (3%) were resistant to chlorfenapyr and this figure progressively increased to approximately 17% in 2002 (BCPC, 2002). The speed with which H armigera larvae developed resistance was due to cross-resistance to high level pyrethroid resistance. Further studies indicated a common esterase mediated resistance mechanism between chlorfenapyr and pyrethroid resistance. Control of Tetrancychus spp on cotton is more effective than that provided by standard miticides and excellent control has been observed with dosages as low as 50 g a.i./ha. The duration of mite control is positively correlated with the application rate. The product does not increase the population in cotton aphids like that observed with pyrethroids.
Chlorfenapyr provided better overall control of insect pests on vegetables such as lettuce, cabbage and broccoli than the commercial standards Bacillus thuringiensis, chlorpyrifos, cypermethrin, diflubenzuron, fenvalerate, methomyl, permethrin and thiodicarb. Vegetable pests such as P.xylostella and L. decemlineata are resistant to conventional insecticides but are controlled by chlorfenpyr.
Chlorfenapyr provides exceptional control of B. phoenicis and Phyllocoptruta oleivora on citrus. Residual control after 60 days has been observed. The addition of spray oil enhances control at lower rates of chlorfenapyr against certain mites. Chlorfenapyr is effective against Lepidoptera species on soybean; it is highly effective on permethrin resistant soybean looper. Chlorfenapyr has shown activity against coffee leafminer at rates of 100-125 g active ingredient but at these rates the control provided was inferior to that obtained with the commercial standards deltamethrin and permethrin.
In laboratory and field studies, the addition of emulsified, petroleum-based oil to chlorfenapyr enhanced the foliar and contact activity of the compound against T.urticae.
Studies of the genetic basis of resistance to the acaricides, chlorfenapyr and etoxazole, which have different chemical structures and modes of action, have been conducted in the two-spotted spider mite, Tetranychus urticae (J. Econ. Entomol, 2002). The resistance ratios calculated from the LC50s of resistant and susceptible strains were 483 for chlorfenapyr and >100.000 for etoxazole. The mode of inheritance of resistance to chlorfenapyr was completely dominant as indicated by mortality caused by the two acaricides in F1 progeny from reciprocal crosses between the resistant and susceptible strains. As shown by the mortality in the F2 progeny, the resistance was under monogenic control for both acaricides. Repeated backcross experiments indicated a linkage relationship among the two acaricide resistances and malate dehydrogenase, although phosphoglucoisomerase was not linked with them. The recombination ratio between the resistances was 14.8%. Heavy spraying of the two acaricides could, therefore, lead to cross-resistance as a consequence of crossing over as the two resistance genes are so close to each other that it would be difficult to segregate them once they came together on the same chromosome. -
CropUseCrop uses:
apples, aubergines, brassica, cabbages, Chinese cabbages, chrysanthemum, cucumbers, citrus, cotton, ground cherries, lettuces, ornamentals, peaches, pears, pepinos, peppers, persimmon, potatoes, tea, tomatilloes, tomatoes, vegetables, vines
Cotton
100-300 g ai/ha
Vegetables
100-360 g ai/ha
Citrus
18.35-62.41 g ai/ha
Ornamentals
180-360 g ai/ha
Greenhouse fruiting vegetables
110-230 g ai/ha
-
PremixMetaflumizone+Chlorfenapyr
Lufenuron+Chlorfenapyr
Ivermectin+Chlorfenapyr
Chlorfenapyr+Tolfenpyrad
Chlorfenapyr+Thiamethoxam
Chlorfenapyr+Pyridaben
Chlorfenapyr+Methoxyfenozide
Chlorfenapyr+Emamectin benzoate
Chlorfenapyr+Clothianidin
Abamectin+Chlorfenapyr
-
Physical PropertiesMolecular weight:407.6; Physical form: White solid. Melting point:100-101 °C; Partition coefficient(n-octanol and water):logP = 4.83; Solubility:Practically insoluble in water. Soluble in acetone, diethyl ether, dimethyl sulfoxide, tetrahydrofuran, acetonitrile, and alcohols.
-
ToxicologyOral:Acute oral LD50 for male rats 441mg/kg , female rats 1152 mg/kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/kg. Moderate eye irritant; non-irritating to skin (rabbits). Inhalation:LC50 for rats 1.9 mg/l air. Phytotoxicity: No phytotoxicity observed at field use rates.?
-
Environmental ProfileEcotoxicology: Bees:LD50 0.2
mg/bee.Birds:Acute oral LD50 for mallard ducks 10 mg/kg, bobwhite quail 34 mg/kg. LC50 (8 d) for mallard ducks 9.4 ppm, bobwhite quail 132 ppm.Daphnia:LC50 (96 h) 6.11 μg/l.Fish:LC50(48 h) for carp 500 μg/l (carp). LC50 (96 h) for rainbow trout 7.44 μg/l, bluegill sunfish 11.6 μg/l.Bee: LD50 0.2 μg/bee.
WATER SOLUBILITY: Practically insoluble.
Mallard duck
LD50 10 mg/kg
Bobwhite quail
LD50 34 mg/kg
Rainbow trout[96 hrs]
LC50 7.44 μg/litre
Bluegill sunfish[96 hrs]
LC50 11.6 μg/litre
Bee[contact]
LD50 0.45 μg/bee
-
Transport InformationSignal Word:CAUTION; Hazard Class:II (Moderately hazardous)
Porduct NewsMore
BASF presents two new insecticides for soybeans and corn in Argentina
Alpha-cypermethrin Acetamiprid Chlorfenapyr
ECHA consults on chlorfenapyr and cyphenothrin as candidates for substitution
Related CompaniesMore
Shanghai Heben-Eastsun Medicaments Co., Ltd.
Country: China
Thiamethoxam Azoxystrobin Boscalid Chlorfenapyr Difenoconazole Fludioxonil Indoxacarb Picoxystrobin Pyraclostrobin Tebuconazole
Xuzhou Jinhe Chemical Co.,Ltd.
Country: China
Thiabendazole Metaldehyde Niclosamide Olamine Tricyclazole Brassinolide Chlorfenapyr Quizalofop-p-ethyl Cyhalofop-butyl LUFENURON Paraquat Dichloride
Jiangsu Weunite Fine Chemical Co., Ltd.
Country: China
Nitenpyram Azoxystrobin Chlorothalonil Acetamiprid Imidaclorprid Bentazone Carbendazim Chlorfenapyr Chlorthal-dimethyl Metaldehyde
Country: China
Imidacloprid Acetamiprid Chlorfenapyr Spirodiclofen Pyraclostrobin Thiamethoxam Prothioconazole CCMP
Country: China
Glufosinate-ammonium 2,4-D MCPA Dicamba Propanil Clethodim Glyphosate Captan Flumioxazin Sulfentrazone

 0
0 Subscribe
Subscribe