-
Common NameTrichlorfon
-
中文通用名敌百虫
-
IUPACdimethyl (RS)-2,2,2-trichloro-1-hydroxyethylphosphonate
or
(RS)-2,2,2-trichloro-1-(dimethoxyphosphinoyl)ethanol -
CASdimethyl (2,2,2-trichloro-1-hydroxyethyl)phosphonate
-
CAS No.52-68-6
-
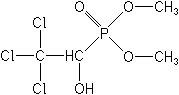
Molecular FormulaC4H8Cl3O4P
-
Molecular Structure
-
Category
-
ActivityInsecticide
-
PremixTrichlorfon+Rotenone
Trichlorfon+malathion
Trichlorfon+fenvalerate
Trichlorfon+beta-cypermethrin
Parathion-methyl+trichlorfon
Parathion+trichlorfon
Diazinon+Trichlorfon
Dusting powder, emulsifiable concentrate, granules, soluble powder, suspension concentrate, wettable powder. Premix Parters: epoxiconazole; tebuconazole; triadimenol;
-
Physical PropertiesMolecular weight:257.4; Physical form:Colourless crystals, with a weak, characteristic odour. Density:1.73 (20 °C); Composition:Racemate, i.e. 1:1 mixture of the (1R)- and (1S)- enantiomers. Melting point:78.5 °C; delayed melting to 84 °C; Vapour pressure:0.21 mPa (20 °C); 0.5 mPa (25 °C); Henry constant:4.4 × 10-7 Pa m3 mol-1 (20 °C); Partition coefficient(n-octanol and water):logP = 0.43 (20 °C); Solubility:In water 120 g/l (20 °C). Readily soluble in common organic solvents (with the exception of aliphatic hydrocarbons and petroleum oils), e.g. hexane 0.1-1, dichloromethane, isopropanol >200, toluene 20-50 (all in g/l, 20 °C).; Stability:Subject to hydrolysis and dehydrochlorination. Decomposition proceeds more rapidly with heating, and above pH 6. Rapidly converted by alkalis to dichlorvos ( q.v.), which is then hydrolysed: DT50;
-
ToxicologyOral:Acute oral LD50 for male and female rats c. 250 mg/kg. Percutaneous:Acute percutaneous LD50 (24 h) for male and female rats >5000 mg/kg. Not irritating to skin and eye (rabbits). Inhalation:LC50 (4 h) for male and female rats >2.3 mg/l air (aerosol). ADI:0.001 mg/kg b.w.
-
Environmental ProfileEcotoxicology:
Algae: ErC50 for Scenedesmus subspicatus >10 mg/l.Bees:Low toxicity to bees and other beneficial insects.Daphnia:LC50 (48 h) 0.00096 mg/l.Fish: LC50 (96 h) for rainbow trout 0.7, golden orfe 0.52 mg/l.
Environmental fate:
EHC 132 notes that trichlorfon is moderately toxic to fish and birds, and moderately to highly toxic to aquatic arthropods, and concludes it should not be sprayed over areas of water.Animals:In animals, trichlorfon is rapidly absorbed and metabolised. Excretion of the radioactivity in the urine is more or less complete within 6 hours. The major metabolites are dimethylphosphoric acid, monomethylphosphoric acid and conjugates of dichloroaceticSoil:Trichlorfon has a relatively high mobility in soil, but it is rapidly metabolised to CO2. Intermediates are dichloroethanol, dichloroacetic acid and trichloroacetic acid. Koc 20 .Plant:In plants, trichlorfon is rapidly hydrolysed. The main metabolites are dimethyl- and monomethyl- phosphoric acid and conjugates of dichloroacetic acid and dichloroethanol. -
Transport InformationHazard Class:II(Moderately hazardous)
Porduct NewsMore
Argentina bans dichlorvos and trichlorfon
Argentina may ban dichlorvos and trichlorfon
Related CompaniesMore
Nantong Jiangshan Agrochemical & Chemicals Limited Liability Co., Ltd.
Country: China
Dichlorvos Trichlorfon Butachlor Acetochlor Alachlor Quizalofop-P-ethyl Prochloraz Prochloraz-manganese Glyphosate

 0
0 Subscribe
Subscribe