-
Common NameChlozolinate
-
中文通用名乙菌利
-
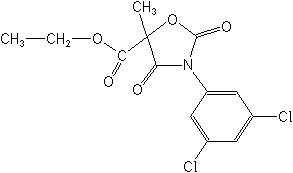
IUPACethyl (RS)-3-(3,5-dichlorophenyl)-5-methyl-2,4-dioxo-1,3-oxazolidine-5-carboxylate
-
CASethyl 3-(3,5-dichlorophenyl)-5-methyl-2,4-dioxo-5-oxazolidinecarboxylate
-
CAS No.84332-86-5
-
Molecular FormulaC13H11Cl2NO5
-
Molecular Structure
-
Category
-
ActivityFungicide
-
Physical PropertiesMolecular weight:332.1; Physical form:Colourless, almost odourless solid. Density:1.441 (20 °C); Melting point:112.6 °C; Vapour pressure:0.013 mPa (25 °C, gas saturation method); Henry constant:2.29×10-3 Pa m3 mol-1 (25 °C, calc.); Partition coefficient(n-octanol and water):logP = 3.15 (22 °C); Solubility:In water c. 2 ppm (25 °C). In acetone, ethyl acetate and 1,2-dichloroethane >250, ethanol c. 13, n-heptane c. 2, xylene c. 60 (all in g/kg, 22 °C).; Stability:Stable to 250 °C (under nitrogen). Stable to light. In solution, hydrolysis occurs at pH 5-9.
-
ToxicologyOral:Acute oral LD50 for rats >4500 mg/kg, mice >10,000 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >5000 mg/kg. Non-irritating to skin and eyes (rabbits). Not a skin sensitiser (guinea pigs). Inhalation:LC50 (4 h) for rats >10 mg/l air.
-
Environmental ProfileEcotoxicology: Algae:EC50 (96 h) for Selenastrum capricornutum 30 mg/l.Bees:LD50 (oral) >100
mg/bee.Birds:Acute oral LD50 for quail and mallard ducks >4500 mg/kg.Daphnia:LC50 (48 h) 1.18 mg/l.Fish: LC50 (96 h) for trout 27.5 mg/l.Other beneficial spp.:Harmless to Phytoseiulus persimilis at up to 2 recommended application rate. Environmental fate: Animals:Chlozolinate is readily absorbed, metabolised and excreted. Metabolites identified in urine of rats are: 3-(3,5-dichlorophenyl)-5-methyloxazolidin-2,4-dione, N-(3,5-dichlorophenyl)-2-hydroxypropionamide, O-1-carboxyethyl-N-3,5-dSoil:In silt-loam, sandy loam and clay loam soils, hydrolysis and decarboxylation occur; aerobic DT50 <7 h.Plant:In plants, chlozolinate undergoes hydrolysis and decarboxylation processes, giving the same metabolites as those identified in animals. -
Transport InformationSignal Word:CAUTION; Hazard Class:III (Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe