-
Common NameEthirimol
-
中文通用名乙嘧酚
-
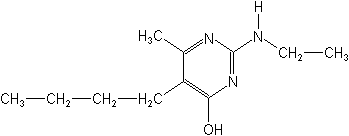
IUPAC5-butyl-2-ethylamino-6-methylpyrimidin-4-ol
-
CAS5-butyl-2-(ethylamino)-6-methyl-4(1H)-pyrimidinone
-
CAS No.23947-60-6
-
Molecular FormulaC11H19N3O
-
Molecular Structure
-
Category
-
ActivityFungicide
-
PremixTrifloxystrobin+Ethirimol
Thiophanate-methyl+Ethirimol
Pyraclostrobin+Ethirimol
Ethirimol+Kresoxim-methyl
Boscalid+Ethirimol
Premix Parters: disulfoton;
-
Physical PropertiesMolecular weight:209.3; Physical form:Colourless crystals. Density:1.21 (25 °C); Composition:Tech. material is 97% pure. Melting point:159-160 °C (phase change at c. 140 °C); Vapour pressure:0.267 mPa (25 °C); Henry constant:>2 × 10-4 ( pH 5.2), >4 × 10-4 ( pH 7.3), >4 × 10-4 ( pH 9.3) Pa m3 mol-1 (20 °C); Partition coefficient(n-octanol and water):logP = 2.3 ( pH 7, 20 °C); pKa:5, weak base (K. Chamberlain et al., Pestic. Sci., 47, 265 (1996) and ref. therein).; Solubility:In water 253 ( pH 5.2), 150 ( pH 7.3), 153 ( pH 9.3) (all in mg/l, 20 °C). In chloroform 150, ethanol 24, acetone 5 (all in g/ kg, 20 °C).; Stability:Stable to heat, and in acidic and alkaline solutions in the dark. Aqueous solution exposed to sunlight and air had DT50 c. 21 d.
-
ToxicologyOral:Acute oral LD50 for female rats 6340, mice 4000, female guinea pigs 500-1000, male rabbits 1000-2000 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats >2000 mg/kg. Mild eye irritant; non-irritating to skin (rats). Not a skin sensitiser (guinea pigs). Inhalation: LC50 for rats >4.92 mg/l. ADI:( JMPR) 0.002 mg/ kg b.w. [1990].
-
Environmental ProfileEcotoxicology:
Bees: LD50 (oral) 1.6 mg/bee.Birds:Acute oral LD50 for hens 4000 mg/ kg.Daphnia: LC50 (48 h) 53 mg/l.Fish: LC50 (96 h) for young brown trout 20, rainbow trout 66 mg/l.
Environmental fate:
Animals:In rats, following oral administration, metabolism involves hydroxylation of the butyl group. Elimination is in the urine.Soil:Soil DT50 14-140 d ( o.m. 1.0-10.1%, pH 7.8-8.1).Plant:In plants, ethirimol undergoes rapid degradation, 2-amino-5-butyl-6-hydroxy-4-methylpyrimidine being found as a metabolite. DT50 in plants c. 3 d. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe