-
Common NameCyclanilide
-
中文通用名环丙酸酰胺
-
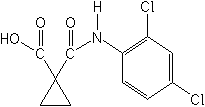
IUPAC1-(2,4-dichloroanilinocarbonyl)cyclopropanecarboxylic acid
-
CAS1-[[(2,4-dichlorophenyl)amino]carbonyl]cyclopropanecarboxylic acid
-
CAS No.113136-77-9
-
Molecular FormulaC11H9Cl2NO3
-
Molecular Structure
-
Category
-
ActivityPlant growth regulator.
Cyclanilide, in mixtures with ethephon, can be used to defoliate cotton, to open mature unopened cotton bolls and to reduce regrowth prior to harvesting. -
CropUseCrop uses:
cotton, rubber
Cyclanilide will only be commercialised in combination products.
-
Premix
Suspension concentrate. Premix Parters: carbendazim triadimenol; chlorpyrifos; imidacloprid; pheromone; piperonyl butoxide propoxur tetramethrin; tebupirimfos.Product
Type
AI concn
Cyclanilide will only be commercialised in combination products
-
Physical PropertiesMolecular weight:274.1; Physical form:White powdery solid. Density:1.47 (20 °C); Melting point:195.5 °C; Vapour pressure:<0.01 mPa (25 °C); 8×10-3 mPa (50 °C); Henry constant:7.41×10-5 Pa m3 mol-1; Partition coefficient(n-octanol and water):logP = 3.25; pKa:3.5 (22 °C); Solubility:Low solubility in water. Soluble in organic solvents.; Stability:Relatively stable to photolysis. Stable to hydrolysis. Decomposes at c. 210 °C. Soluble in organic solvents.
-
ToxicologyOral:Acute oral LD50 for female rats 208 mg/kg, male rats 315 mg/kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/kg. Slight skin irritant. Inhalation:LC50 (4 h) for rats >5.15 mg/l.
-
Environmental ProfileEcotoxicology:
Algae:Moderately toxic.Bees:LD50 (contact) for bees >100 μg/bee.Birds:Acute oral LD50 for bobwhite quail 216 mg/kg, mallard ducks >215 mg/kg b.w. Dietary LC50 (8 d) for bobwhite quail 2849, mallard ducks 1240 ppm. Daphnia:EC50 (48 h) >13 mg/l.Fish:LC50 (96 h) for bluegill sunfish >16 mg/l, rainbow trout >11 mg/l, sheepshead minnow 49 mg/l.Worms:Moderately toxic. Environmental fate:
Animals:Rapidly excreted, primarily as unchanged cyclanilide.Soil:Low to moderate persistence, DT50 c. 16 d under aerobic conditions. Degrades primarily by microbial activity. Medium to low mobility (average Koc 346). Consequently little potential for leachPlant:Little degradation occurs in plants; cyclanilide is the major residue.
Mallard duck
LD50 >215 mg/kg
Rainbow trout [96 hrs]
LC50 >11 mg/litre
Bluegill sunfish [96 hrs]
LC50 >16 mg/litre
Earthworm [14 days]
LC50 469 mg/kg
Bee [contact]
LD50 >100 μg/bee
Bee [oral]
LD50 89.5 μg/bee
Daphnia magna
EC50 13 mg/L
Alga (S carpricornutum)
EC50 (cell density) 1.7 mg/L
Fate in soil:
The DT50 for soil photolysis is 151 days at 25°C.
Cyclanilide is degraded by microbes in the soil. In aerobic soil metabolism studies, the major metabolite is 2,4-dichloroaniline (2,4-DCA) at 1.5 - 16% of the applied radioactivity.
The DT50 for laboratory aerobic soil metabolism is 15-49 days (mean = 29 days) at 20°C. The corresponding DT90 is 50-162 days (mean = 96 days).
Under laboratory anaerobic conditions, the DT50 for cyclanilide at 20°C is 15 months or more.
In field dissipation studies in France and Spain, the DT50 for cyclanilide is 11-45 days (mean = 28 days) and the DT90 was 37-151 days (mean = 94 days). In the US, the DT50 was measured at 34-114 days (mean = 65 days).
The Koc for cyclanilide ranges from 194 (loamy sand) to 565 (silty loam), with Kd ranging from 1.05 - 3.83.
Cyclanilide has low to moderate soil persistance and mobility.Fate in aquatic systems:
Cyclanilide is primarily degraded by microbes in water.
It is not hydrolysed at pH 5,7 or 9. The product does not bioaccumulate in water.
The DT50 for aqueous photostability ranges from 997 hours (pH 5, 9) to 1173 hours (pH 7).
In water / sediment studies, the DT50 for cyclanilide is 17-18 days (water layer) and 56-63 days (whole system). After 105 days, 14-18% of the parent compound is found in the water phase and 13-15% in the soil layer. -
Transport InformationSignal Word:DANGER; Hazard Class:Ib(Highly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe