-
Common NameEndothal
-
中文通用名茵多酸
-
IUPAC7-oxabicyclo[2.2.1]heptane-2,3-dicarboxylic acid
-
CAS7-oxabicyclo[2.2.1]heptane-2,3-dicarboxylic acid
-
CAS No.145-73-3
-
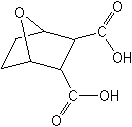
Molecular FormulaC8H10O5
-
Molecular Structure
-
Category
-
ActivityHerbicide, algicide, plant growth regulator
-
Physical PropertiesMolecular weight:186.2; Physical form:Colourless crystals (monohydrate). Density:1.431 at 20 °C; Composition:Of the 4 theoretical stereoisomers of endothal, the rel-(1R,2S,3R,4S)- isomer is the most effective herbicide (US 2550494). Melting point:144 °C (monohydrate); Flash point:Non-flammable; Vapour pressure:2.09 ×10-5 mPa (24.3 °C); Henry constant:3.8 ×10-13 Pa m3 mol-1 (calc.); Partition coefficient(n-octanol and water):logP = -2.09 (unstated pH); pKa:A dibasic acid ( pKa1 3.4, pKa2 6.7); Solubility:In water 100 g/ kg (20 °C). In methanol 280, dioxane 76, acetone 70, isopropanol 17, diethyl ether 1, benzene 0.1 (all in g/ kg, 20 °C).; Stability:Stable to light. Stable up to c. 90 °C, above which it undergoes a slow conversion to the anhydride. Endothal is a dibasic acid, and forms water-soluble amine and alkali-metal salts.;
-
ToxicologyOral:Acute oral LD50 for rats 38-54 mg/kg (acid), 206 mg/ kg (66.7% formulation of the amine salt). Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/l (acid). Inhalation: LC50(14 d) 0.68 mg/l (acid). ADI:( JMPR) 0.006 mg/kg b.w. [1998].
-
Environmental ProfileEcotoxicology:
Algae:Toxic to algae.Bees:Not toxic to bees.Birds:Acute LD50 for ducks 111 mg/kg b.w.Daphnia: LC50 (48 h) 92 mg/l.Fish: LC50 (96 h) for trout 49, bluegill sunfish 77 mg/l.
Environmental fate:
Animals:Rapidly absorbed. Elimination DT50 1.8-2.5 h.Soil: DT50 in aerobic soil 8.5 d. Kd 1.3-37.1. For a review, see Simsiman et al. (Residue Rev., 1976, 62, 131-174).Plant:Residues in the plant were mainly endothal. -
Transport InformationHazard Class:O (Obsolete as pesticide, not classified)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe