-
Common NameFenbutatin oxide
-
中文通用名苯丁锡
-
IUPACbis[tris(2-methyl-2-phenylpropyl)tin] oxide
-
CAShexakis(2-methyl-2-phenylpropyl)distannoxane
-
CAS No.13356-08-6
-
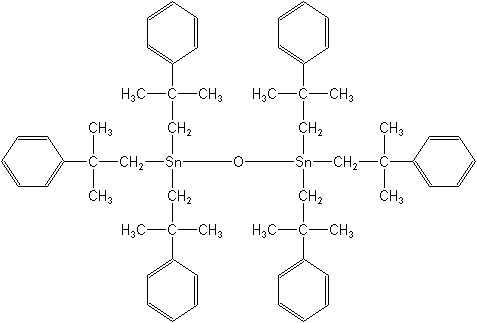
Molecular FormulaC60H78OSn2
-
Molecular Structure
-
Category
-
ActivityAcaricide
-
PremixSpirotetramat+Fenbutatin oxide
Spirodiclofen+Fenbutatin oxide
-
Physical PropertiesMolecular weight:1052.7; Physical form:Colourless crystals. Density:1290-1330 kg/m3 (20 °C); Melting point:138-139 °C ( tech.); Flash point:Not autoflammable, but tech. will explode in a dust cloud if ignited; Vapour pressure:8.5 × 10-5 mPa (extrapolated to 20 °C); Henry constant:1.79 × 10-2 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 5.2; Solubility:In water 0.005 mg/l (23 °C). In acetone 6, benzene 140, dichloromethane 380 (all in g/l, 23 °C). Very slightly soluble in aliphatic hydrocarbons and mineral oils.; Stability:Extremely stable to heat, light, and atmospheric oxygen. Water causes conversion of fenbutatin oxide to tris(2-methyl-2-phenylpropyl)tin hydroxide, which is reconverted to the parent compound slowly at room temperature and rapidly at 98 °C.;
-
ToxicologyOral:Acute oral LD50 for rats 2631, mice 1450, dogs >1500 mg/ kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000, rats >1000 mg/kg. Irritating to skin and severely irritating to eyes. Inhalation: LC50 0.23 mg a.i. (as dust)/l. Phytotoxicity:Phytotoxic to tangerines, tangelos, and some varieties of grapefruit. Otherwise, non-phytotoxic when used as directed. ADI:( JMPR) 0.03 mg/ kg b.w. [1997].
-
Environmental ProfileEcotoxicology:
Bees:Acute oral LD50 >0.1 mg/bee.Birds:Dietary LC50 (8 d) for bobwhite quail 5065 mg/ kg diet.Daphnia: LC50 (24 h) 0.05-0.08 mg/l.Fish: LC50 (48 h) for rainbow trout 0.27 mg a.i./l (as WP formulation).Other beneficial spp.:No adverse effects to a wide range of predatory and parasitic arthropods.
Environmental fate:
Soil:In soil, it is metabolised to dihydroxy-bis(2-methyl-2-phenylpropyl)stannane and 2-methyl-2-phenylpropyl stannonic acid, presumably ultimately forming tin oxide/hydroxide. In tests in commercial use, there was minimal movement of fenbutatin oxide or its m -
Transport InformationHazard Class:III(Slightly hazardous)
Porduct NewsMore
EU withdraws approval of acaricide fenbutatin oxide
Related CompaniesMore
Shanghai Heben-Eastsun Medicaments Co., Ltd.
Country: China
Thiamethoxam Azoxystrobin Boscalid Chlorfenapyr Difenoconazole Fludioxonil Indoxacarb Picoxystrobin Pyraclostrobin Tebuconazole
Country: China
Clethodim Hexazinone Mesotrione Abamectin Acetamiprid Dimethoate Fipronil Imidacloprid Fosetyl-aluminium 2,4-D

 0
0 Subscribe
Subscribe