-
Common NameHeptachlor
-
中文通用名七氯
-
IUPAC1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro-4,7-methanoindene
-
CAS1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro-4,7-methano-1H-indene
-
CAS No.76-44-8
-
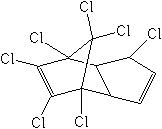
Molecular FormulaC10H5Cl7
-
Molecular Structure
-
Category
-
ActivityInsecticide
-
Physical PropertiesMolecular weight:373.3g/mol; Physical form:Crystalline solid; ( tech., waxy solid). Density:1.58 (20 ℃) ( tech.); Composition:Tech. grade is c. 72% heptachlor and 28% related compounds. Melting point:95-96℃; ( tech. 55-60℃); Vapour pressure:53 mPa (25℃, pure); Henry constant:3.53 × 102 Pa m3 mol-1 ( calc.); Solubility:In water 0.056 mg/l (25-29℃). Soluble in many organic solvents, e.g. in acetone 75, benzene 106, xylene 102, cyclohexanone 119, carbon tetrachloride 113, ethanol 4.5 (all in g/100 ml). Tech. in cyclohexanone 1.65 kg/l, ; Stability:Stable in daylight, air, moisture, and moderate heat (up to 160℃). Not readily dehydrochlorinated, but is susceptible to epoxidation.
-
ToxicologyOral:Acute oral LD50 for rats 147-220, guinea pigs 116, mice 68 mg/ kg. Percutaneous:Acute percutaneous LD50 for rabbits 200-2000, rats 119-250 mg/kg. Irritating to eyes; not a skin irritant (rabbits). Inhalation: LC50 (4 h) for rats exposed to heptachlor in an aerosol >2.0 but <200 mg/l air.
-
Environmental ProfileEcotoxicology:
Birds:Acute oral LD50 for mallard ducklings >2000 mg/kg. Dietary LC50 (8 d) for bobwhite quail 450-700, Japanese quail 80-95, pheasants 250-275 mg/ kg dietFish: LC50 (96 h) for rainbow trout 7, bluegill sunfish 26, fathead minnow 78-130 μg/l.?
Environmental fate:
Animals:The principal metabolites in animals are heptachlor epoxide (found in the tissues, faeces, and urine), and 1-exo-hydroxychlordene epoxide, a hydrophilic metabolite excreted in the urine. There is a strong tendency for the epoxide to accumulate inSoil:In water, heptachlor rapidly undergoes hydrolysis to 1-hydroxychlordene, which then undergoes microbial epoxidation to 1-hydroxy-2,3-epoxychlordene. Similar degradation pathways occur in moist soil. The half-life in soil is 9-10 months when used at agricuPlant:In plants, heptachlor epoxide is a metabolite. -
Transport InformationSignal Word:WARNING; Hazard Class:II(Moderately hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe