-
Common NameInabenfide
-
中文通用名抗倒胺
-
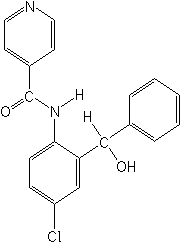
IUPAC(RS)-4′-chloro-2′-(α-hydroxybenzyl)isonicotinanilide
-
CASN-[4-chloro-2-(hydroxyphenylmethyl)phenyl]-4-pyridinecarboxamide
-
CAS No.82211-24-3
-
Molecular FormulaC19H15ClN2O2
-
Molecular Structure
-
Category
-
ActivityPlant Growth Regulator
-
Physical PropertiesMolecular weight:338.8; Physical form:Pale yellow-brown or colourless crystals. Melting point:210-212 °C; Vapour pressure:0.063 mPa (20 °C); Partition coefficient(n-octanol and water):logP = 3.13; Solubility:In water 1 mg/l (30 °C). In acetone 3.6, ethyl acetate 1.43, xylene 0.58, methanol 2.35, ethanol 1.61, chloroform 0.59, dimethylformamide 6.72, acetonitrile 0.58, tetrahydrofuran 1.61, hexane 8 ×10-4 (all ; Stability:Stable to heat and sunlight. Slightly unstable in alkaline media; after 2 weeks at 40 °C, hydrolysed 16.2% ( pH 2), 49.5% ( pH 5), 83.9% ( pH 7), 100% ( pH 11).
-
ToxicologyOral:Acute oral LD50 for rats and mice >15 000 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats and mice >5000 mg/kg. Not a skin or eye irritant to rabbits, nor skin sensitiser to guinea pigs. Inhalation: LC50 (4 h) for rats >0.46 mg/l air. Phytotoxicity:Non-phytotoxic to rice.
-
Environmental ProfileEcotoxicology:
Daphnia: LC50 (3 h) >30 mg/l.Fish: LC50 (48 h) for carp >30, grey mullet 11 mg/l.
Environmental fate:
Animals:The major urinary metabolite in the rat is 4-hydroxyinabenfide (H. Kinoshita et al., Xenobiotica, 1987, 17, 925).Soil:Half-life under Japanese paddy field conditions, c. 4 months.Plant:Metabolised to inabenfide ketone. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide

 0
0 Subscribe
Subscribe