-
Common NameMetolachlor
-
中文通用名异丙甲草胺
-
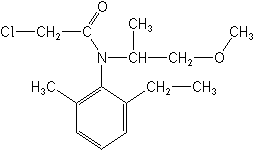
IUPAC2-chloro-N-(6-ethyl-o-tolyl)-N-[(1RS)-2-methoxy-1-methylethyl]acetamide
or
(aRS,1RS)-2-chloro-6′-ethyl-N-(2-methoxy-1-methylethyl)acet-o-toluidide -
CAS2-chloro-N-(2-ethyl-6-methylphenyl)-N-(2-methoxy-1-methylethyl)acetamide
-
CAS No.51218-45-2
-
Molecular FormulaC15H22ClNO2
-
Molecular Structure
-
Category
-
ActivityHerbicide
-
PremixSulfentrazone+S-metolachlor
S-metolachlor+Terbuthylazine
Pendimethalin+Metolachlor
Oxyfluorfen+S-metolachlor
Nicosulfuron+S-metolachlor+Atrazine
Metribuzin+Metolachlor+2,4-D-ethylhexyl
Metolachlor+Terbutryn
Metolachlor+metribuzin
Metolachlor+fomesafen
Metolachlor+Atrazine
Bensulfuron-methyl+metolachlor
Atrazine+S-metolachlor
Atrazine+mesotrione+S-metolachlor
Atrazine Bicyclopyrone Mesotrione S-metolachlor
Emulsifiable concentrate, granules.
Premix Parters: flufenacet;
-
Physical PropertiesMolecular weight:283.8; Physical form:Colourless to light tan liquid. Density:1.12 (20 °C); Composition:Racemic mixture of (1S)- and (1R)- isomers. Melting point:-62.1 °C; Flash point:190 °C (1013 mbar); Vapour pressure:4.2 mPa (25 °C) (OECD 104); Henry constant:2.4 ×10-3 Pa m3 mol-1 ( calc.); Partition coefficient(n-octanol and water):logP = 2.9 (25 °C); Solubility:In water 488 mg/l (25 °C) ( OECD 105). Miscible with benzene, toluene, ethanol, acetone, xylene, hexane, dimethylformamide, ethylene dichloride, cyclohexanone, methanol, octanol, and dichloromethane. Insoluble in ethylene ; Stability:Stable up to c. 275 °C. Hydrolysed by strong alkalis and strong mineral acids. On hydrolysis in buffer (20 °C), DT50 ( calc.) >200 d (pH >10).;
-
ToxicologyOral:Acute oral LD50 for rats 2780 mg/ kg. Percutaneous:Acute percutaneous LD50 for rats >3170 mg/kg. Mild skin and eye irritant (rabbits). May cause skin sensitisation (guinea pigs). Inhalation: LC50 (4 h) for rats >1.75 mg/l air. Phytotoxicity:Well tolerated by most broad-leaved crops, maize, sorghum (safened with fluxofenim or oxabetrinil). ADI:0.008 mg/ kg b.w.
-
Environmental ProfileEcotoxicology:
Algae:EC50 for Scenedesmus subspicatus 0.1 mg/l.Bees: LD50 (oral and contact) >110 mg/bee.Birds:Acute oral LD50 for mallard ducks and bobwhite quail >2150 mg/kg. Dietary LC50 (8 d) for bobwhite quail and mallard ducks >10 000 mg/ kg.Daphnia:LC50 (48 h) 25 mg/l.Fish:LC50 (96 h) for rainbow trout 3.9, carp 4.9, bluegill sunfish 10 mg/l.Worms:LC50 (14 d) for earthworms 140 mg/ kg soil.
Environmental fate:
Animals:Rapidly oxidised by rat liver microsomal oxygenases via dechlorination, O-demethylation and side-chain oxidation (J. Agric. Food Chem., 1989, 37, 1088).Soil:Major aerobic metabolites are derivatives of oxanilic and sulfonic acids. DT50 in soil c. 20 d (field). Koc 121-309.Plant:In plants, metabolism involves natural product conjugation of the chloroacetyl group, and hydrolysis and sugar conjugation at the ether group. Final metabolites are polar, water-soluble, and non-volatile. -
Transport InformationHazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Related CompaniesMore
Hangzhou Qingfeng Imp.& Exp. Co.,Ltd.
Country: China
S-metolachlor Fomesafen Flumioxazin Clethodim Imazapyr Tebuthiuron Azoxystrobin Tebuconazole Oxyfluorfen Metolachlor
Jiangsu Yongan Chemical Co., Ltd.
Country: China
Pendimethalin Methoxyfenozide 2,4-D Clethodim Quinclorac Glufosinate-ammonium Metolachlor Atrazine Pendimethalin
Country: China
Oxyfluorfen Tebuconazole Metolachlor Azoxystrobin Clopyralid Triclopyr Tebuthiuron Acifluorfen Clethodim Imazapyr Picloram Pretilachlor Bifenthrin Amitraz Mesotrione
Xingfa(Shanghai)International Trading Co.,Ltd.
Country: China
Glyphosate Glufosinate Chlorpyrifos Paraquat 2,4-D Clethodim Atrazine Azoxystrobin Lambda-cyhalothrin Metolachlor
Shenzhen Baocheng Chemical Industry CO.,LTD.
Country: China
Glyphosate Emamectin benzoate Lambda-cyhalothrin Pyraclostrobin Bromacil Glufosinate-ammonium Tembotrione Prothioconazole Topramezone Chlorantraniliprole

 0
0 Subscribe
Subscribe