-
Common NameTriadimenol
-
中文通用名三唑醇
-
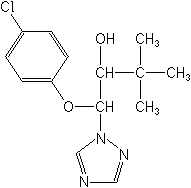
IUPAC(1RS,2RS;1RS,2SR)-1-(4-chlorophenoxy)-3,3-dimethyl-1-(1H-1,2,4-triazol-1-yl)butan-2-ol
-
CASβ-(4-chlorophenoxy)-α-(1,1-dimethylethyl)-1H-1,2,4-triazole-1-ethanol
-
CAS No.55219-65-3
-
Molecular FormulaC14H18ClN3O2
-
Molecular Structure
-
Category
-
ActivityFungicide
-
PremixTrifloxystrobin+metalaxyl+triadimenol
TRIADIMENOL+TRIFLUMURON
Kresoxim-methyl+anthraquinone+fenpropimorph+triadimenol+triazoxide
CYPERMETHRIN+TRIADIMENOL
Emulsifiable concentrate, flowable, granule, seed dressing, water dispersible granule, water-oil emulsion, wettable powder. Premix Parters: butafenacil; dicamba; isoproturon;
-
Physical PropertiesMolecular weight:295.8; Physical form:Colourless crystals, with a weak characteristic odour. Density:A: 1.237; B: 1.299 (22 °C); Composition:(1RS,2SR) is referred to here as diastereoisomer A; (1RS,2RS) is referred to here as diastereoisomer B. The ratio of A:B is c. 7:3. Melting point:A: 138.2 °C; B: 133.5 °C; eutectic A+B: 110 °C; ( tech., 103-120 °C); Vapour pressure:A: 6 × 10-4 mPa; B: 4 × 10-4 mPa (20 °C); Henry constant:A: 3 × 10-6; B: 4 × 10-6 (Pa m3 mol-1, 20 °C); Partition coefficient(n-octanol and water):A: logP = 3.08; B: logP = 3.28 (25 °C); Solubility:In water A: 62; B: 33 mg/l (20 °C). In dichloromethane 200-500, isopropanol 50-100, hexane 0.1-1.0, toluene 20-50 (all in g/l, 20 °C).; Stability:Both diastereoisomers are stable to hydrolysis; DT50 (20 °C) >1 y ( pH 4, 7 or 9).;
-
ToxicologyOral:Acute oral LD50 for rats c. 700, mice c. 1300 mg/kg. Percutaneous:Acute percutaneous LD50 for rats >5000 mg/kg. Non-irritating to eyes and skin (rabbits). Inhalation: LC50 (4 h) for rats >0.9 mg/l air (aerosol). ADI:( JMPR) 0.03 mg/kg b.w. [1985].
-
Environmental ProfileEcotoxicology:
Algae: ErC50 for Scenedesmus subspicatus 3.7 mg/l.Bees:Non-toxic to honeybees.Birds:Acute oral LD50 for bobwhite quail >2000 mg/kg.Daphnia:LC50 (48 h) 51 mg/l.Fish:LC50 (96 h) for golden orfe 17.4-27.3, rainbow trout 14-23.5, bluegill sunfish 15 mg/l.Worms:LC50 for Eisenia foetida 772 mg/kg dry soil.
Environmental fate:
Animals:In rats, triadimenol was metabolised mainly by oxidation of the tert-butyl moiety to the corresponding alcohol and then to carboxylic acid. A small fraction of these compounds was conjugated. Oxidation at the hydroxyl group to the corresponding kSoil:In soil, triadimenol is a degradation product of triadimefon ( q.v.). Degradation involving hydrolytic cleavage leads to the formation of 4-chlorophenol. Metabolism of the individual triadimenol enantiomers proceeds at different rPlant:The most important breakdown reactions of triadimenol in plants after seed treatment and spray application are conjugation of the a.i. with various sugar compounds (especially hexose) and oxidation at the tert-butyl moiety. The res -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Colombia’s Constitutional Court bans chlorpyrifos
ADAMA Canada moves forward with lambda-cyhalothrin sales for 2023
Mexico: Import of glyphosate drops from 16,500 tons to 8,200 tons
Glyphosate prices drop 30% in Argentina
One year extension of EU glyphosate approval failed to pass in the latest meeting
Related CompaniesMore
Jiangsu Sword Agrochemicals Co., Ltd.
Country: China
Triadimefon Triadimenol Tebuconazole Diniconazole Bitertanol Cyproconazole Propineb Paclobutrazol Metribuzin Bentazone
Yancheng Limin Chemical Co.,Ltd
Country: China
Triadimefon Triadimenol Hexaconazole Flutriafol Imidacloprid Paclobutrazol Tebuconazole Thifluzamide Indoxacarb Uniconazole

 0
0 Subscribe
Subscribe