-
Common NameCyhalothrin
-
中文通用名氯氟氰菊酯
-
IUPAC(RS)-α-cyano-3-phenoxybenzyl (1RS,3RS)-3-[(Z)-2-chloro-3,3,3-trifluoropropenyl]-2,2-dimethylcyclopropanecarboxylate
or
(RS)-α-cyano-3-phenoxybenzyl (1RS)-cis-3-[(Z)-2-chloro-3,3,3-trifluoropropenyl]-2,2-dimethylcyclopropanecarboxylate -
CAScyano(3-phenoxyphenyl)methyl (1R,3R)-rel-3-[(1Z)-2-chloro-3,3,3-trifluoro-1-propenyl]-2,2-dimethylcyclopropanecarboxylate
-
CAS No.68085-85-8
-
Molecular FormulaC23H19ClF3NO3
-
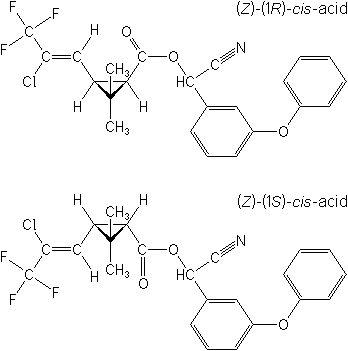
Molecular Structure
-
Category
-
Activity
Cyhalothrin is a pesticide to be used against animal, public, and agricultural pests including beetles, aphids, and butterfly larvae
-
CropUseWheat, Barley, Beans (dry) include Butter beans, Cow peas, Kidney beans, Lentil, Lima beans, Pegia, Sultani, Sultapyar and White beans, Potato, Sugar beet
-
Pest Spectrumbeetles, aphids, and butterfly larvae
-
FormulationWP = Wettable powder
-
PremixThiamethoxam+lambda-cyhalothrin
Spinosad+lambda-cyhalothrin
Pymetrozine+Lambda-cyhalothrin
Propoxur+Lambda-cyhalothrin
Profenofos+lambda-cyhalothrin
Lambda-cyhalothrin+triazophos
Lambda-cyhalothrin+thiosultap-monosodium
Lambda-cyhalothrin+Pirimiphos-methyl
Lambda-cyhalothrin+pirimicarb
Lambda-cyhalothrin+Piperonyl butoxide+tetramethrin
Lambda-cyhalothrin+methomyl+phoxim
Lambda-cyhalothrin+methomyl
Lambda-cyhalothrin+malathion+phoxim
Lambda-cyhalothrin+malathion
Lambda-cyhalothrin+Lufenuron
Lambda-cyhalothrin+isocarbophos
Lambda-cyhalothrin+Hexaflumuron
Lambda-cyhalothrin+fenitrothion
Lambda-cyhalothrin+endosulfan+phoxim
Lambda-cyhalothrin+endosulfan
Lambda-cyhalothrin+emamectin benzoate
Lambda-cyhalothrin+Dinotefuran
Lambda-cyhalothrin+dimethoate
Lambda-cyhalothrin+diazinon
Lambda-cyhalothrin+chlorpyrifos-methyl
Lambda-cyhalothrin+chlorpyrifos+Piperonyl butoxide
Lambda-cyhalothrin+chlorpyrifos
Lambda-cyhalothrin+chlorantraniliprole
Lambda-cyhalothrin+buprofezin
Imidacloprid+lambda-cyhalothrin
Endosulfan+lambda-cyhalothrin
Diflubenzuron+Lambda-cyhalothrin
Diflubenzuron+Lambda-cyhalothrin
Dichlorvos+lambda-cyhalothrin
Diafenthiuron+lambda-cyhalothrin
Cyhalothrin+parathion-methyl+isocarbophos
Clothianidin+Lambda-Cyhalothrin
Acetamiprid+lambda-cyhalothrin
Abamectin+lambda-cyhalothrin
-
Physical PropertiesMolecular weight:449.9; Physical form:Yellow to brown viscous oil ( tech.). Density:1.25 (25 °C); Composition:Tech. grade cyhalothrin has purity 90% m/m, of which 95% is cis- isomers. Flash point:204 °C ( tech., Pensky-Martens closed cup); Vapour pressure:0.001 mPa (20 °C, est.); Partition coefficient(n-octanol and water):logP = 6.9 (20 °C); pKa:>9 (hydrolysis prevents measurement); Solubility:In water 0.005 mg/l ( pH 6.5, 20 °C). In acetone, dichloromethane, methanol, diethyl ether, ethyl acetate, hexane, toluene, all >500 g/l (20 °C).; Stability:Stable to decomposition and cis/trans isomerisation for at least 4 years in the dark at 50 °C. Stable to light; loss on storage in the light is <10% in 20 months. Decomposes at 275 °C. Slowly hydrolysed by water in sunlight.
-
ToxicologyOral:Acute oral
LD50 for male rats 166, female rats 114, guinea pigs >5000, rabbits >1000 mg/kg. Percutaneous:Acute percutaneous LD50 for male rats 1000-2500, female rats 200-2500, rabbits >2500 mg/kg. Moderate eye irritant; not a skin irritant (rabbits). Moderate skin sensitiser (guinea pigs). Inhalation: LC50 (4 h) for rats >0.086 mg/l. -
Environmental ProfileEcotoxicology:
Birds:Acute oralLD50 for mallard ducks >5000 mg/kg.Daphnia: LC50 (48 h) 0.38 μg/l.Fish: LC50 (96 h) for rainbow trout 0.00054 mg/l.
Environmental fate:
Animals:In rats, following oral administration, cyhalothrin is rapidly eliminated in urine and faeces. The ester group is hydrolysed, both moieties forming polar conjugates.Soil:In soil,DT50 c. 4-12 weeks. Leaching of cyhalothrin and its degradation products through a range of soil types is negligible. -
Transport InformationHazard Class:II (Moderately hazardous)
Porduct NewsMore
Jiangsu Yangnong Chemical to tackle illegal Cyhalothrin in China

 0
0 Subscribe
Subscribe