-
Common NameTau-fluvalinate
-
中文通用名氟胺氰菊酯
-
IUPAC(RS)-α-cyano-3-phenoxybenzyl N-(2-chloro-α,α,α-trifluoro-p-tolyl)-D-valinate
-
CAScyano(3-phenoxyphenyl)methyl N-[2-chloro-4-(trifluoromethyl)phenyl]-D-valinate
-
CAS No.102851-06-9
-
Molecular FormulaC26H22ClF3N2O3
-
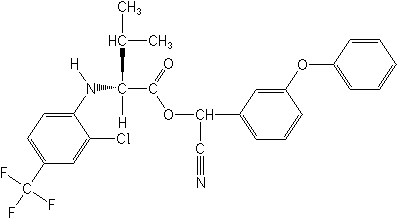
Molecular Structure
-
Category
-
ActivityAcaricide/Miticide
Insecticide
-
Physical PropertiesMolecular weight:502.9; Physical form:Viscous amber oil, with a moderate or weak sweetish odour (tech.). Density:1.262 (25 °C); Composition:Material is a 1:1 mixture of (R)-a-cyano-, 2-(R)- and (S)-a-cyano-, 2-(R)- diastereoisomers. Flash point:90 °C ( tech.; Pensky-Martens closed cup); Vapour pressure:9 × 10-8 mPa; Henry constant:4.04 × 10-5 Pa m3 mol-1 (calc.); Partition coefficient(n-octanol and water):logP = 4.26 (25 °C); Solubility:In water 1.03 ppb ( pH 7, 20 °C). Soluble in toluene, acetonitrile, isopropanol, dimethylformamide, n-octanol; in iso-octane 108 g/l.; Stability:Tech. material is stable for 2 years at room temperature (20-28 °C). Undergoes decomposition on exposure to sunlight; DT50 9.3-10.7 min (aqueous solution buffered pH 5), c. 1 d (thin fi;
-
ToxicologyOral:Acute oral LD50 for female rats 261, male rats 282 mg a.i. (in corn oil)/kg. Percutaneous:Acute percutaneous LD50 for rabbits >2000 mg/kg; slight skin irritant, mild eye irritant (rabbits). Inhalation: LC50 (4 h) for rats >0.56 mg/l air (as 240 g/l EC formulation).
-
Environmental ProfileEcotoxicology:
Algae:LC50 for Scenedesmus subspicatus >2.2 mg/l.Bees:Not hazardous to bees at recommended doses; LD50 (24 h, contact topical) 6.7 µg tech./bee; (ingestion) 163 µg tech./bee.Birds:Acute oral LD50 for bobwhite quail >2510 mg/kg. Dietary LC50 (8 d) for bobwhite quail and mallard ducks >5620 mg/kg diet.Daphnia: LC50 (48 h) 0.001 mg/l.Fish: LC50 (96 h) for bluegill sunfish 0.0062, rainbow trout 0.0027, carp 0.0048 mg/l.Worms: LC50 (14 d) >1000 ppm.Other beneficial spp.:Generally safe to moderately toxic to beneficials, except spiders, predatory mites, some lady beetles and some predatory bugs.
Environmental fate:
Animals:In rats, following oral administration, c. 90% is excreted within 4 d, of which 20-40% is in the urine and 60-80% in the faeces. The principal metabolites are "anilino acid", 3-phenoxybenzoic acid (3-PBA) and 4'-3-PBA as faecal metabolites, and 4Soil:In soil under aerobic conditions (lab.), DT50 12-92 d. Primary metabolites include the corresponding anilino acids and the parent aniline. Koc(adsorption) >110 000, (desorption) >39 000.Plant:The parent compound itself accounts for >90% of the residue following treatment with tau-fluvalinate. Minor residues include decarboxyfluvalinate, "anilino acid", haloaniline, 3-PBA and 4'-hydroxy-3-PBA. DT50 2-6 w. -
Transport InformationSignal Word:CAUTION; Hazard Class:III(Slightly hazardous)
Porduct NewsMore
Orthosulfamuron boosts sugarcane production, study finds
Glyphosate price plummets 40% in one year in Argentina
Indian govt stops imports of herbicide Glufosinate priced below Rs 1,289 per kg
Carbendazim fungicide wins victory in Brazilian Parliament
Corteva presents new pre-emergent herbicide Linear for sugarcane in Brazil
Picloram Triclopyr Aminopyralid
Revolutionizing disease prevention: BASF launches new rice fungicide Cevya® in China
Thiamethoxam is allowed again in Brazil by a judicial decision
Bayer develops alternative to glyphosate herbicide
Related CompaniesMore
Country: China

 0
0 Subscribe
Subscribe